CHEMICAL IDENTIFICATION
-
RTECS NUMBER :
-
MA8050000
-
CHEMICAL NAME :
-
Glycerol
-
CAS REGISTRY NUMBER :
-
56-81-5
-
BEILSTEIN REFERENCE NO. :
-
0635685
-
LAST UPDATED :
-
199712
-
DATA ITEMS CITED :
-
50
-
MOLECULAR FORMULA :
-
C3-H8-O3
-
MOLECULAR WEIGHT :
-
92.11
-
WISWESSER LINE NOTATION :
-
Q1YQ1Q
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
-
TYPE OF TEST :
-
Standard Draize test
-
ROUTE OF EXPOSURE :
-
Administration onto the skin
-
SPECIES OBSERVED :
-
Rodent - rabbit
-
TYPE OF TEST :
-
Standard Draize test
-
ROUTE OF EXPOSURE :
-
Administration into the eye
-
SPECIES OBSERVED :
-
Rodent - rabbit
-
TYPE OF TEST :
-
Standard Draize test
-
ROUTE OF EXPOSURE :
-
Administration into the eye
-
SPECIES OBSERVED :
-
Rodent - rabbit
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human
-
DOSE/DURATION :
-
1428 mg/kg
-
TOXIC EFFECTS :
-
Behavioral - headache Gastrointestinal - nausea or vomiting
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
12600 mg/kg
-
TOXIC EFFECTS :
-
Behavioral - general anesthetic Behavioral - muscle weakness Liver - other changes
-
TYPE OF TEST :
-
LC50 - Lethal concentration, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Inhalation
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
>570 mg/m3/1H
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Intraperitoneal
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
4420 mg/kg
-
TOXIC EFFECTS :
-
Behavioral - toxic psychosis Cardiac - other changes Kidney, Ureter, Bladder - other changes
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Subcutaneous
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
100 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Intravenous
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
5566 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - mouse
-
DOSE/DURATION :
-
4090 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Intraperitoneal
-
SPECIES OBSERVED :
-
Rodent - mouse
-
DOSE/DURATION :
-
8700 mg/kg
-
TOXIC EFFECTS :
-
Behavioral - altered sleep time (including change in righting reflex)
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Subcutaneous
-
SPECIES OBSERVED :
-
Rodent - mouse
-
DOSE/DURATION :
-
91 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Intravenous
-
SPECIES OBSERVED :
-
Rodent - mouse
-
DOSE/DURATION :
-
4250 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - rabbit
-
DOSE/DURATION :
-
27 gm/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Administration onto the skin
-
SPECIES OBSERVED :
-
Rodent - rabbit
-
DOSE/DURATION :
-
>10 gm/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Intravenous
-
SPECIES OBSERVED :
-
Rodent - rabbit
-
DOSE/DURATION :
-
53 gm/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - guinea pig
-
DOSE/DURATION :
-
7750 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
16800 mg/kg/28D-C
-
TOXIC EFFECTS :
-
Endocrine - changes in adrenal weight
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
96 gm/kg/30D-I
-
TOXIC EFFECTS :
-
Blood - changes in leukocyte (WBC) count Blood - changes in serum composition (e.g. TP, bilirubin, cholesterol) Biochemical - Enzyme inhibition, induction, or change in blood or tissue levels - true cholinesterase
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - mouse
-
DOSE/DURATION :
-
560 gm/kg/8W-C
-
TOXIC EFFECTS :
-
Lungs, Thorax, or Respiration - structural or functional change in trachea or bronchi
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
100 mg/kg
-
SEX/DURATION :
-
male 1 day(s) pre-mating
-
TOXIC EFFECTS :
-
Reproductive - Fertility - post-implantation mortality (e.g. dead and/or resorbed implants per total number of implants)
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Intratesticular
-
DOSE :
-
280 mg/kg
-
SEX/DURATION :
-
male 2 day(s) pre-mating
-
TOXIC EFFECTS :
-
Reproductive - Paternal Effects - spermatogenesis (incl. genetic material, sperm morphology, motility, and count) Reproductive - Paternal Effects - testes, epididymis, sperm duct
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Intratesticular
-
DOSE :
-
1600 mg/kg
-
SEX/DURATION :
-
male 1 day(s) pre-mating
-
TOXIC EFFECTS :
-
Reproductive - Fertility - male fertility index (e.g. # males impregnating females per # males exposed to fertile nonpregnant females)
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Intratesticular
-
DOSE :
-
862 mg/kg
-
SEX/DURATION :
-
male 1 day(s) pre-mating
-
TOXIC EFFECTS :
-
Reproductive - Paternal Effects - spermatogenesis (incl. genetic material, sperm morphology, motility, and count)
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Intratesticular
-
DOSE :
-
119 mg/kg
-
SEX/DURATION :
-
male 1 day(s) pre-mating
-
TOXIC EFFECTS :
-
Reproductive - Paternal Effects - spermatogenesis (incl. genetic material, sperm morphology, motility, and count) Reproductive - Paternal Effects - testes, epididymis, sperm duct
MUTATION DATA
-
TEST SYSTEM :
-
Rodent - rat
-
DOSE/DURATION :
-
1 gm/kg
-
REFERENCE :
-
TGANAK Tsitologiya i Genetika. Cytology and Genetics. For English translation, see CYGEDX. (V/O Mezhdunarodnaya Kniga, 113095 Moscow, USSR) V.1- 1967- Volume(issue)/page/year: 19,436,1985 *** REVIEWS *** ACGIH TLV-TWA 10 mg/m3, Inhalable particulate DTLVS* The Threshold Limit Values (TLVs) and Biological Exposure Indices (BEIs) booklet issues by American Conference of Governmental Industrial Hygienists (ACGIH), Cincinnati, OH, 1996 Volume(issue)/page/year: TLV/BEI,1997 TOXICOLOGY REVIEW INMEAF Industrial Medicine. (Chicago, IL) V.1-18, 1932-49. For publisher information, see IOHSA5. Volume(issue)/page/year: 9,-,1940 *** U.S. STANDARDS AND REGULATIONS *** EPA FIFRA 1988 PESTICIDE SUBJECT TO REGISTRATION OR RE-REGISTRATION FEREAC Federal Register. (U.S. Government Printing Office, Supt. of Documents, Washington, DC 20402) V.1- 1936- Volume(issue)/page/year: 54,7740,1989 MSHA STANDARD:NUISANCE PARTICULATES (mist) DTLWS* "Documentation of the Threshold Limit Values for Substances in Workroom Air," Supplements. For publisher information, see 85INA8. Volume(issue)/page/year: 3,20,1973 OSHA PEL (Gen Indu):8H TWA 15 mg/m3, total dust CFRGBR Code of Federal Regulations. (U.S. Government Printing Office, Supt. of Documents, Washington, DC 20402) Volume(issue)/page/year: 29,1910.1000,1994 OSHA PEL (Gen Indu):8H TWA 5 mg/m3, respirable fraction CFRGBR Code of Federal Regulations. (U.S. Government Printing Office, Supt. of Documents, Washington, DC 20402) Volume(issue)/page/year: 29,1910.1000,1994 OSHA PEL (Construc):8H TWA 15 mg/m3, total dust CFRGBR Code of Federal Regulations. (U.S. Government Printing Office, Supt. of Documents, Washington, DC 20402) Volume(issue)/page/year: 29,1926.55,1994 OSHA PEL (Construc):8H TWA 5 mg/m3, respirable fraction CFRGBR Code of Federal Regulations. (U.S. Government Printing Office, Supt. of Documents, Washington, DC 20402) Volume(issue)/page/year: 29,1926.55,1994 OSHA PEL (Shipyard):8H TWA 15 mg/m3, total dust CFRGBR Code of Federal Regulations. (U.S. Government Printing Office, Supt. of Documents, Washington, DC 20402) Volume(issue)/page/year: 29,1915.1000,1993 OSHA PEL (Shipyard):8H TWA 5 mg/m3, respirable fraction CFRGBR Code of Federal Regulations. (U.S. Government Printing Office, Supt. of Documents, Washington, DC 20402) Volume(issue)/page/year: 29,1915.1000,1993 *** OCCUPATIONAL EXPOSURE LIMITS *** OEL-AUSTRALIA:TWA 10 mg/m3 JAN 1993 OEL-BELGIUM:TWA 10 mg/m3 JAN 1993 OEL-FINLAND:TWA 20 mg/m3 JAN 1993 OEL-FRANCE:TWA 10 mg/m3 JAN 1993 OEL-THE NETHERLANDS:TWA 10 mg/m3 JAN 1993 OEL-UNITED KINGDOM:TWA 10 mg/m3 JAN 1993 OEL IN BULGARIA, COLOMBIA, JORDAN, KOREA check ACGIH TLV OEL IN NEW ZEALAND, SINGAPORE, VIETNAM check ACGIH TLV *** NIOSH STANDARDS DEVELOPMENT AND SURVEILLANCE DATA *** NIOSH OCCUPATIONAL EXPOSURE SURVEY DATA : NOHS - National Occupational Hazard Survey (1974) NOHS Hazard Code - 35085 No. of Facilities: 86657 (estimated) No. of Industries: 358 No. of Occupations: 198 No. of Employees: 1085329 (estimated) NOES - National Occupational Exposure Survey (1983) NOES Hazard Code - 35085 No. of Facilities: 67054 (estimated) No. of Industries: 310 No. of Occupations: 215 No. of Employees: 2135546 (estimated) No. of Female Employees: 1346631 (estimated)
|
 CAS#:107-18-6
CAS#:107-18-6 CAS#:50-99-7
CAS#:50-99-7 CAS#:57-48-7
CAS#:57-48-7 CAS#:50-70-4
CAS#:50-70-4 CAS#:201230-82-2
CAS#:201230-82-2 CAS#:100-79-8
CAS#:100-79-8 CAS#:67-56-1
CAS#:67-56-1 CAS#:122-32-7
CAS#:122-32-7 CAS#:58-86-6
CAS#:58-86-6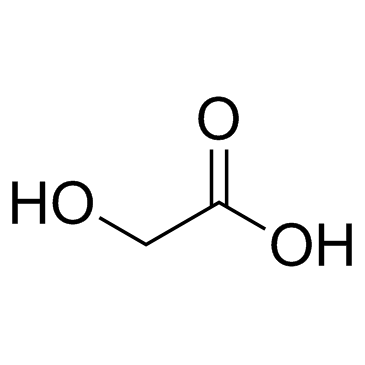 CAS#:79-14-1
CAS#:79-14-1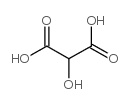 CAS#:80-69-3
CAS#:80-69-3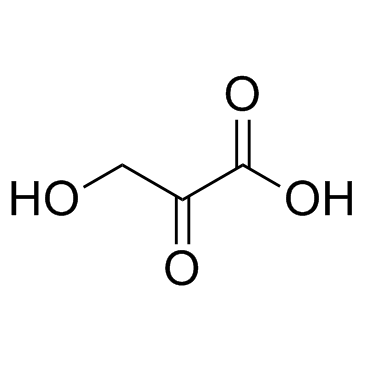 CAS#:1113-60-6
CAS#:1113-60-6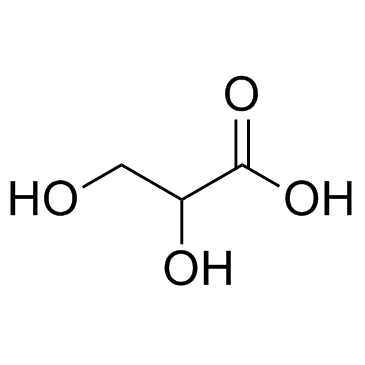 CAS#:473-81-4
CAS#:473-81-4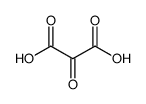 CAS#:473-90-5
CAS#:473-90-5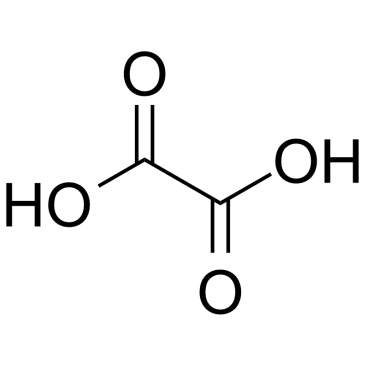 CAS#:144-62-7
CAS#:144-62-7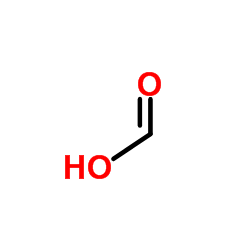 CAS#:64-18-6
CAS#:64-18-6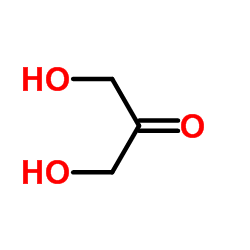 CAS#:96-26-4
CAS#:96-26-4 CAS#:298-12-4
CAS#:298-12-4 CAS#:110-85-0
CAS#:110-85-0
