溴乙烯
一般危化品
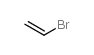
溴乙烯结构式
|
常用名 | 溴乙烯 | 英文名 | bromoethene |
|---|---|---|---|---|
| CAS号 | 593-60-2 | 分子量 | 106.94900 | |
| 密度 | 1.517 g/mL at 25 °C(lit.) | 沸点 | 16 °C750 mm Hg(lit.) | |
| 分子式 | C2H3Br | 熔点 | −139 °C(lit.) | |
| MSDS | 中文版 美版 | 闪点 | 1 °F | |
| 符号 |



GHS02, GHS07, GHS08 |
信号词 | Danger |
|
Reactivity in nucleophilic vinylic substitution (S(N)V):S(N)Vπ versus S(N)Vσ mechanistic dichotomy.
J. Org. Chem. 78(17) , 8574-84, (2013) The intrinsic electronic factors that determine reactivity in prototypical identity nucleophilic vinylic substitution reactions, X(-) + ViX → XVi + X(-) (Vi = vinyl), have been studied by performing quantum chemical calculations (OPBE/6-311++G(d,p)). Of the t... |
|
|
Asymmetric synthesis of enantioenriched (+)-elaeokanine A.
J. Org. Chem. 71(15) , 5674-8, (2006) The key transformation in the total synthesis of (+)-elaeokanine A was accomplished by asymmetric deprotonation of N-Boc pyrrolidine, followed by the reaction of the in situ generated enantioenriched stereogenic cuprate reagent with (E)-4-bromo-1-iodo-1-trime... |
|
|
Formation of enehydrazine intermediates through coupling of phenylhydrazines with vinyl halides: entry into the Fischer indole synthesis.
Angew. Chem. Int. Ed. Engl. 52(4) , 1266-9, (2013)
|
|
|
Total synthesis of polycavernoside A, a lethal toxin of the red alga Polycavernosa tsudai.
J. Org. Chem. 70(14) , 5449-60, (2005) [structure: see text] Two approaches to the synthesis of the aglycon 120 of polycavernoside A (1) were developed, only one of which was completed. The successful "second-generation" route assembled the aglycon seco acids 102 and 106 via Nozaki-Hiyama-Kishi co... |
|
|
Synthesis of the C1-C13 tetraenoate subunit of the chivosazoles.
Org. Lett. 12(16) , 3724-7, (2010) Using a combination of asymmetric vinylogous Mukaiyama aldol and Stille cross-coupling reactions, an advanced polyene fragment of the chivosazoles was prepared in a highly stereocontrolled manner. This key C1-C13 pentaene subunit, featuring the conjugated (2E... |
|
|
New one-pot synthesis of (E)-beta-aryl vinyl halides from styrenes.
Org. Lett. 11(15) , 3390-3, (2009) A new, efficient protocol for the highly stereoselective one-pot synthesis of (E)-beta-aryl vinyl iodides and (E)-beta-aryl vinyl bromides from styrenes based on sequential ruthenium-catalyzed silylative coupling-N-halosuccinimide-mediated halodesilylation re... |
|
|
Diastereoselective additions of chiral vinylzinc reagents to alpha-chiral aldehydes.
Org. Lett. 6(3) , 445-8, (2004) [reaction: see text] Additions of vinylic zinc bromide reagents to alpha-chiral aldehydes (R(1) = CH(2)OTBS, R(2) = Me; R(1) = Me, R(2) = OTBS) in the presence of lithiated (+)- or (-)-N-methylephedrine proceed with predominant reagent control to afford anti ... |
|
|
CuI-catalyzed intramolecular O-vinylation of carbonyl compounds.
Chem. Commun. (Camb.) (28) , 3574-6, (2005) The first copper-catalyzed intramolecular O-vinylation of carbonyl compounds with vinyl bromides was reported, among which the efficient formation of 5-, 6- and even 7-membered cyclic alkenyl ethers was achieved with beta-ketoesters as nucleophiles. |
|
|
Iron-catalysed, hydride-mediated reductive cross-coupling of vinyl halides and Grignard reagents.
Chem. Commun. (Camb.) 48(10) , 1580-2, (2012) An iron-catalysed, hydride-mediated reductive cross-coupling reaction has been developed for the preparation of alkanes. Using a bench-stable iron(II) pre-catalyst, reductive cross-coupling of vinyl iodides, bromides and chlorides with aryl- and alkyl Grignar... |
|
|
A new reactivity pattern for vinyl bromides: cine-substitution via palladium catalysed C-N coupling/Michael addition reactions.
Org. Biomol. Chem. 3(17) , 3094-5, (2005) Palladium catalysed C-N bond formation can be used to convert vinyl bromides to the corresponding enamines, which are reacted in situ with alkylidene malonates to provide Michael adducts. The overall transformation results in cine-substitution of the starting... |