tryptamine
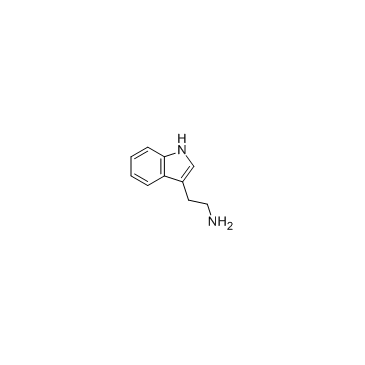
tryptamine structure
|
Common Name | tryptamine | ||
|---|---|---|---|---|
| CAS Number | 61-54-1 | Molecular Weight | 160.216 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 378.8±0.0 °C at 760 mmHg | |
| Molecular Formula | C10H12N2 | Melting Point | 113-116 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 187.7±8.1 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of tryptamineTryptamine is a monoamine alkaloid, similar to other trace amines, is believed to play a role as a neuromodulator or neurotransmitter. |
| Name | tryptamine |
|---|---|
| Synonym | More Synonyms |
| Description | Tryptamine is a monoamine alkaloid, similar to other trace amines, is believed to play a role as a neuromodulator or neurotransmitter. |
|---|---|
| Related Catalog | |
| Target |
Human Endogenous Metabolite |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 378.8±0.0 °C at 760 mmHg |
| Melting Point | 113-116 °C(lit.) |
| Molecular Formula | C10H12N2 |
| Molecular Weight | 160.216 |
| Flash Point | 187.7±8.1 °C |
| Exact Mass | 160.100052 |
| PSA | 41.81000 |
| LogP | 1.38 |
| Vapour Pressure | 0.0±0.8 mmHg at 25°C |
| Index of Refraction | 1.669 |
| Water Solubility | negligible |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
| Hazard Codes | Xi |
| Risk Phrases | 20/21/22-36/37/38-41-37/38-22 |
| Safety Phrases | S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | NL4020000 |
| HS Code | 29339990 |
| Precursor 9 | |
|---|---|
| DownStream 9 | |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Use of Commercial Dry Yeast Products Rich in Mannoproteins for White and Rosé Sparkling Wine Elaboration.
J. Agric. Food Chem. 63 , 5670-81, (2015) In sparkling wines, mannoproteins released during yeast autolysis largely affect their final quality. This process is very slow and may take several months. The aim of this work was to study the effec... |
|
|
Enantioselective formal synthesis of ent-rhynchophylline and ent-isorhynchophylline.
Chem. Commun. (Camb.) 49(19) , 1954-6, (2013) Starting from (S)-tryptophanol, a formal synthesis of ent-rhynchophylline and ent-isorhynchophylline, involving stereoselective cyclocondensation, spirocyclization, and alkylation reactions, and the f... |
|
|
A separation of tyramine on a 2-(4-methoxyphenyl)ethylamine imprinted polymer: an answer from theoretical and experimental studies.
Talanta 129 , 155-64, (2014) A 2-(4-methoxyphenyl)ethylamine imprinted polymer (MIP) was successfully applied for the selective separation of tyramine. A computational analysis was used to predict the affinity of the polymer matr... |
| 2-(1H-Indol-3-yl)ethylamine |
| Tryptamine |
| 1H-Indole-3-ethanamine |
| 2-indol-3-yl-ethylamine |
| Indole-ethylamine |
| L-TRYPTAMINE |
| EINECS 200-510-5 |
| 2-Indol-3-yl-aethylamin |
| Tryptamin |
| 3-Indoleethanamine |
| 2-(1H-Indol-3-yl)ethanamine |
| MFCD00005661 |
| TRYPTAMINE(P) |
| T56 BMJ D2Z |
| [2-(1H-indol-3-yl)-ethyl]amine |
| TRIPTAMINE |
| (1H-indol-3-yl)ethanamine |
| 2-indol-3-ylethylamine |
 CAS#:31731-23-4
CAS#:31731-23-4 CAS#:103549-24-2
CAS#:103549-24-2 CAS#:771-51-7
CAS#:771-51-7 CAS#:1042701-07-4
CAS#:1042701-07-4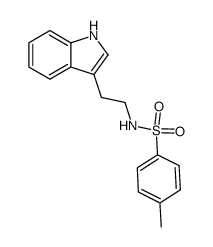 CAS#:86658-78-8
CAS#:86658-78-8 CAS#:73-22-3
CAS#:73-22-3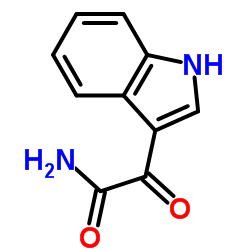 CAS#:5548-10-7
CAS#:5548-10-7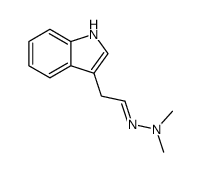 CAS#:74321-36-1
CAS#:74321-36-1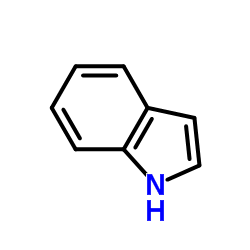 CAS#:120-72-9
CAS#:120-72-9![1-(trifluoromethyl)-9H-pyrido[3,4-b]indole structure](https://image.chemsrc.com/caspic/151/112037-79-3.png) CAS#:112037-79-3
CAS#:112037-79-3![5,5-diethyl-6-[2-(1H-indol-3-yl)ethylamino]pyrimidine-2,4-dione structure](https://image.chemsrc.com/caspic/360/105891-76-7.png) CAS#:105891-76-7
CAS#:105891-76-7![2,2,2-trifluoro-N-[2-(1-methylindol-3-yl)ethyl]acetamide structure](https://image.chemsrc.com/caspic/363/111217-41-5.png) CAS#:111217-41-5
CAS#:111217-41-5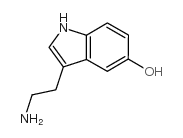 CAS#:50-67-9
CAS#:50-67-9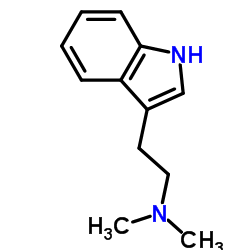 CAS#:61-50-7
CAS#:61-50-7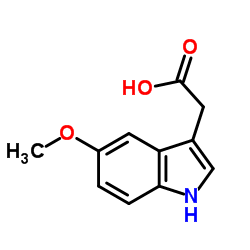 CAS#:608-07-1
CAS#:608-07-1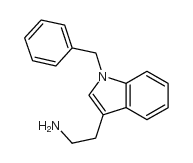 CAS#:4307-98-6
CAS#:4307-98-6![1-phenyl-2,3,4,9-tetrahydro-1H-pyrido[3,4-b]indole structure](https://image.chemsrc.com/caspic/484/3790-45-2.png) CAS#:3790-45-2
CAS#:3790-45-2 CAS#:16765-79-0
CAS#:16765-79-0
