1,2,4-Trihydroxybenzene
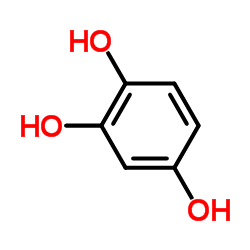
1,2,4-Trihydroxybenzene structure
|
Common Name | 1,2,4-Trihydroxybenzene | ||
|---|---|---|---|---|
| CAS Number | 533-73-3 | Molecular Weight | 126.110 | |
| Density | 1.5±0.1 g/cm3 | Boiling Point | 334.5±12.0 °C at 760 mmHg | |
| Molecular Formula | C6H6O3 | Melting Point | 140 °C (subl.)(lit.) | |
| MSDS | Chinese USA | Flash Point | 176.9±14.2 °C | |
| Symbol |


GHS05, GHS07 |
Signal Word | Danger | |
Use of 1,2,4-Trihydroxybenzene1,2,4-Trihydroxybenzene (Hydroxyhydroquinone), a by-product of coffee bean roasting, increases intracellular Ca2+ concentration in rat thymic lymphocytes[1]. |
| Name | benzene-1,2,4-triol |
|---|---|
| Synonym | More Synonyms |
| Description | 1,2,4-Trihydroxybenzene (Hydroxyhydroquinone), a by-product of coffee bean roasting, increases intracellular Ca2+ concentration in rat thymic lymphocytes[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.5±0.1 g/cm3 |
|---|---|
| Boiling Point | 334.5±12.0 °C at 760 mmHg |
| Melting Point | 140 °C (subl.)(lit.) |
| Molecular Formula | C6H6O3 |
| Molecular Weight | 126.110 |
| Flash Point | 176.9±14.2 °C |
| Exact Mass | 126.031693 |
| PSA | 60.69000 |
| LogP | 0.06 |
| Vapour Pressure | 0.0±0.8 mmHg at 25°C |
| Index of Refraction | 1.677 |
| InChIKey | GGNQRNBDZQJCCN-UHFFFAOYSA-N |
| SMILES | Oc1ccc(O)c(O)c1 |
| Water Solubility | freely soluble |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |


GHS05, GHS07 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H302-H315-H318-H335 |
| Precautionary Statements | P261-P280-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn:Harmful; |
| Risk Phrases | R20/21/22;R36/37/38 |
| Safety Phrases | S26-S39-S36/37/39-S22 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | DC4200000 |
| HS Code | 2907299090 |
| Precursor 7 | |
|---|---|
| DownStream 10 | |
| HS Code | 2907299090 |
|---|---|
| Summary | 2907299090 polyphenols; phenol-alcohols。supervision conditions:AB(certificate of inspection for goods inward,certificate of inspection for goods outward)。VAT:17.0%。tax rebate rate:9.0%。MFN tariff:5.5%。general tariff:30.0% |
|
A convenient screening method to differentiate phenolic skin whitening tyrosinase inhibitors from leukoderma-inducing phenols.
J. Dermatol. Sci. 80 , 18-24, (2015) Tyrosinase is able to oxidize a great number of phenols and catechols to form ortho-quinones. Ortho-quinones are highly reactive compounds that exert cytotoxicity through binding with thiol enzymes an... |
|
|
Degradation of sulfonamide antibiotics by Microbacterium sp. strain BR1 - elucidating the downstream pathway.
New Biotechnology 32 , 710-5, (2015) Microbacterium sp. strain BR1 is among the first bacterial isolates which were proven to degrade sulfonamide antibiotics. The degradation is initiated by an ipso-substitution, initiating the decay of ... |
|
|
The TetR-type transcriptional repressor RolR from Corynebacterium glutamicum regulates resorcinol catabolism by binding to a unique operator, rolO.
Appl. Environ. Microbiol. 78(17) , 6009-16, (2012) The rol (designated for resorcinol) gene cluster rolRHMD is involved in resorcinol catabolism in Corynebacterium glutamicum, and RolR is the TetR-type regulator. In this study, we investigated how Rol... |
| 4-Hydroxycatechol |
| 1,2,4-Trihydroxybenzene |
| 1,2,4-Benzenetriol |
| 1,3,4-Benzenetriol |
| 1,2,3-Trihydroxybenzene |
| benzene-1,2,3-triol |
| EINECS 208-575-1 |
| Hydroxyhydroquinone |
| Benzene-1,2,4-triol |
| Benzol-1,2,3-triol |
| 1,2,3-Benzenetriol |
| gallol |
| 4-06-00-07338 (Beilstein Handbook Reference) |
| MFCD00002198 |
 CAS#:613-03-6
CAS#:613-03-6 CAS#:95-77-2
CAS#:95-77-2 CAS#:615-67-8
CAS#:615-67-8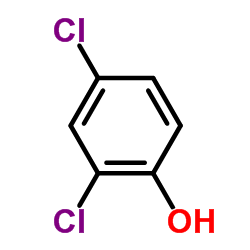 CAS#:120-83-2
CAS#:120-83-2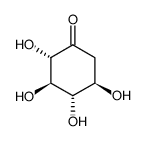 CAS#:78963-40-3
CAS#:78963-40-3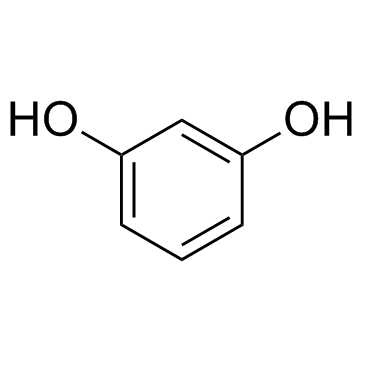 CAS#:108-46-3
CAS#:108-46-3 CAS#:112473-07-1
CAS#:112473-07-1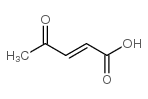 CAS#:4743-82-2
CAS#:4743-82-2 CAS#:3569-82-2
CAS#:3569-82-2 CAS#:529-84-0
CAS#:529-84-0 CAS#:55939-28-1
CAS#:55939-28-1 CAS#:504-02-9
CAS#:504-02-9 CAS#:4281-40-7
CAS#:4281-40-7 CAS#:123-31-9
CAS#:123-31-9![Spiro[isobenzofuran-1(3H),9'-[9H]xanthen]-3-one,2',3',6',7'-tetrahydroxy- structure](https://image.chemsrc.com/caspic/272/2320-44-7.png) CAS#:2320-44-7
CAS#:2320-44-7 CAS#:106-50-3
CAS#:106-50-3 CAS#:88404-14-2
CAS#:88404-14-2
