Tyramine

Tyramine structure
|
Common Name | Tyramine | ||
|---|---|---|---|---|
| CAS Number | 51-67-2 | Molecular Weight | 137.179 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 275.1±23.0 °C at 760 mmHg | |
| Molecular Formula | C8H11NO | Melting Point | 160-162 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 141.3±13.3 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of TyramineTyramine is an indirectly acting sympathomimetic amine that releases norepinephrine from presynaptic nerve terminals. |
| Name | tyramine |
|---|---|
| Synonym | More Synonyms |
| Description | Tyramine is an indirectly acting sympathomimetic amine that releases norepinephrine from presynaptic nerve terminals. |
|---|---|
| Related Catalog | |
| Target |
Human Endogenous Metabolite |
| In Vitro | Tyramine is an indirectly acting sympathomimetic amine that releases norepinephrine from presynaptic nerve terminals[1]. Tyramine potentiates the postsynaptic inhibitory effects of norepinephrine on the firing of cortical neurones in the rat[2]. Although tyramine exists in three isomerie forms, the orthoisomer and metaisomers are present in very low concentrations and it is the paraisomer that is implied when reference is made to tyramine. The tyramines are generally present in nanogram/gram concentrations in brain tissue, but they have a very rapid turnover. The CNS role of tyramine is unclear, it has been suggested that it is involved in the modulation of dopamine synthesis and may also regulate norepinephrine turnover[3]. |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 275.1±23.0 °C at 760 mmHg |
| Melting Point | 160-162 °C(lit.) |
| Molecular Formula | C8H11NO |
| Molecular Weight | 137.179 |
| Flash Point | 141.3±13.3 °C |
| Exact Mass | 137.084061 |
| PSA | 46.25000 |
| LogP | 1.38 |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.600 |
| Storage condition | Refrigerator, Under Inert Atmosphere |
| Stability | Stable. Incompatible with strong acids, strong oxidizing agents. |
| Water Solubility | 1g/95mL (15 ºC) |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi:Irritant |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26-S36-S37/39 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | SJ5950000 |
| HS Code | 2922299090 |
| Precursor 9 | |
|---|---|
| DownStream 10 | |
| HS Code | 2922299090 |
|---|---|
| Summary | 2922299090. other amino-naphthols and other amino-phenols, other than those containing more than one kind of oxygen function, their ethers and esters; salts thereof. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Regulators of autophagosome formation in Drosophila muscles.
PLoS Genet. 11(2) , e1005006, (2015) Given the diversity of autophagy targets and regulation, it is important to characterize autophagy in various cell types and conditions. We used a primary myocyte cell culture system to assay the role... |
|
|
Use of Commercial Dry Yeast Products Rich in Mannoproteins for White and Rosé Sparkling Wine Elaboration.
J. Agric. Food Chem. 63 , 5670-81, (2015) In sparkling wines, mannoproteins released during yeast autolysis largely affect their final quality. This process is very slow and may take several months. The aim of this work was to study the effec... |
|
|
Mixed alkyl aryl phosphonate esters as quenched fluorescent activity-based probes for serine proteases.
Org. Biomol. Chem. 13(8) , 2293-9, (2015) Activity-based probes (ABPs) are powerful tools for the analysis of active enzyme species in whole proteomes, cells or animals. Quenched fluorescent ABPs (qABPs) can be applied for real time imaging, ... |
| Phenol, 4-(ethylamino)- |
| Uteramine |
| Tyramin |
| MFCD00008193 |
| 4-Ethylaminophenol |
| Tocosine |
| 4-(2-aminoethyl)phenol |
| 4-(2-aminoethyl)-pheno |
| p-Hydroxyphenylethylamine |
| Tyrosamine |
| Systogene |
| 4-Hydroxyphenethylamine |
| p-Tyramine |
| 4-(Ethylamino)phenol |
| 4-(Ethylamino)benzolol |
| 2-(4-Hydroxyphenyl)ethylamine |
| EINECS 200-115-8 |
| 4-hydroxy-phenethylamine |
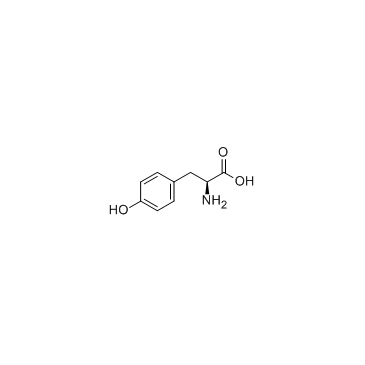 CAS#:60-18-4
CAS#:60-18-4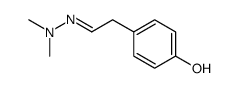 CAS#:74321-28-1
CAS#:74321-28-1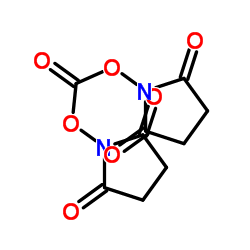 CAS#:74124-79-1
CAS#:74124-79-1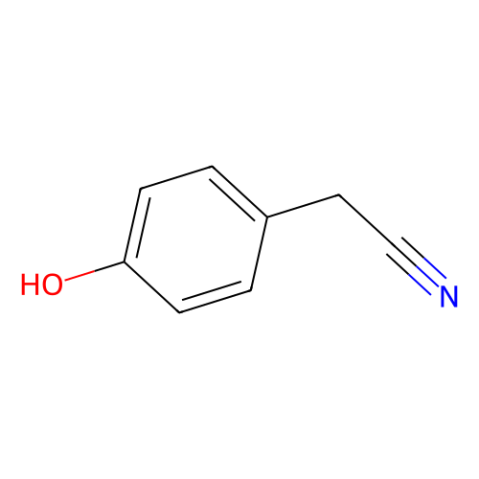 CAS#:14191-95-8
CAS#:14191-95-8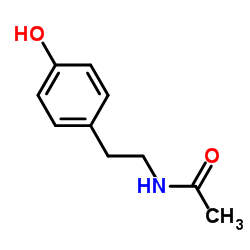 CAS#:1202-66-0
CAS#:1202-66-0![N-{2-[4-(tetrahydro-2H-pyran-2-yloxy)phenyl]ethyl}formamide Structure](https://www.chemsrc.com/caspic/449/1521270-08-5.png) CAS#:1521270-08-5
CAS#:1521270-08-5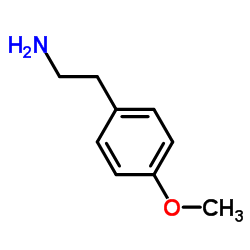 CAS#:55-81-2
CAS#:55-81-2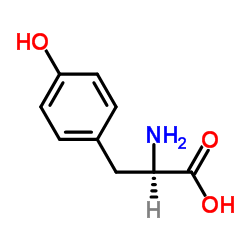 CAS#:556-02-5
CAS#:556-02-5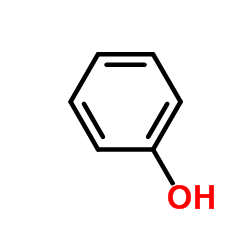 CAS#:108-95-2
CAS#:108-95-2![5-[[2-(4-hydroxyphenyl)ethylamino]methyl]-2-methoxy-phenol structure](https://www.chemsrc.com/caspic/221/4579-60-6.png) CAS#:4579-60-6
CAS#:4579-60-6 CAS#:30798-64-2
CAS#:30798-64-2 CAS#:14383-56-3
CAS#:14383-56-3 CAS#:20284-85-9
CAS#:20284-85-9 CAS#:4442-85-7
CAS#:4442-85-7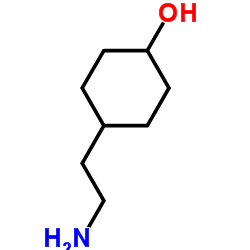 CAS#:148356-06-3
CAS#:148356-06-3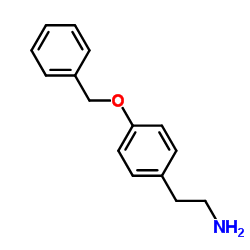 CAS#:51179-05-6
CAS#:51179-05-6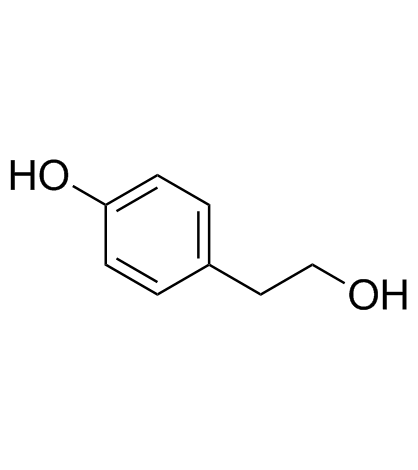 CAS#:501-94-0
CAS#:501-94-0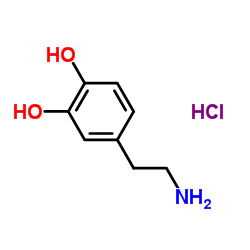 CAS#:51-61-6
CAS#:51-61-6
