442-51-3
| Name | harmine |
|---|---|
| Synonyms |
Yajeine
Banisterine Harmine 7-Methoxy-1-methyl-9H-β-carboline Telepathin Yagein Garmin Telepathien Harmin Yageine 7-methoxy-1-methyl-9H-pyrido[3,4-b]indole EINECS 207-131-4 Telepathine BANISTERINE MONOHYDRATE 7-Methoxy-1-methyl-9H-pyrido(3,4-b)indole MFCD00150055 |
| Description | Harmine is a natural dual-specificity tyrosine phosphorylation-regulated kinase ((DYRK)) inhibitor with anticancer and anti-inflammatory activities. |
|---|---|
| Related Catalog | |
| Target |
5-HT2A Receptor:397 nM (Ki) DYRK1A RAD51 |
| In Vitro | Harmine is an inhibitor of 5-HT2A, with an Ki of 397 nM[1]. Harmine inhibits tau phosphorylation by DYRK1A by selected DANDYs, with an IC50 of 190 nM[2].Harmine negatively regulates HR by interfering Rad51 recruitment, resulting in severe cytotoxicity in hepatoma cells. Furthermore, NHEJ inhibitor Nu7441 markedly sensitizes Hep3B cells to the anti-proliferative effects of Harmine[3]. |
| In Vivo | It is shown that brain water content is significantly increased in the TBI group. Treatment with Harmine significantly reduces the tissue water content at 1, 3 and 5 days, compared with the TBI group. Harmine treatment significantly reduces the escape latency at 3 and 5 days, compared with the TBI group. Post-TBI administration of Harmine significantly improves the motor function recovery of the rats at 1, 3 and 5 days following TBI, compared with the TBI group without Harmine treatment. The neuronal survival rate in the Harmine-treated group is significantly increased, compared with the TBI group. Administration of Harmine results in marked elevation in the expression of GLT-1, compared with the TBI group. The administration of Harmine significantly reduces the expression of caspase 3, compared with the TBI group[4]. |
| Animal Admin | Rats[4] A total of 150 male Sprague-Dawley rats (age, 10-12 weeks; weighing, 280-320 g; are used in the present study. The rats are randomly divided into three groups: Sham-operated group (sham; n=15); the TBI group (TBI; n=35) and the TBI + Harmine-treated group (Harmine; n=35). Harmine is administered immediately following TBI (i.p, 30 mg/kg per day) for up to 5 days. The sham and TBI groups receive equal volumes of 0.9% saline solution (i.p.). The rats are grouped as follows for examination of behavioral recovery: Sham, n=3; TBI, n=7; and Harmine, n=7. Following TBI, the NSS is evaluated at 1, 3 and 5 days. Each rat is assessed by an observer who is blinded to the animal treatment[4]. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 421.4±40.0 °C at 760 mmHg |
| Melting Point | 262-264 °C(lit.) |
| Molecular Formula | C13H12N2O |
| Molecular Weight | 212.247 |
| Flash Point | 139.8±17.0 °C |
| Exact Mass | 212.094955 |
| PSA | 37.91000 |
| LogP | 3.17 |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.706 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Hazard Codes | Xn:Harmful |
|---|---|
| Risk Phrases | R20/21/22;R36 |
| Safety Phrases | S22-S24/25-S36/37-S26 |
| RIDADR | 1544 |
| WGK Germany | 3 |
| RTECS | UV0175000 |
| Packaging Group | III |
| Hazard Class | 6.1 |
| HS Code | 2933990090 |
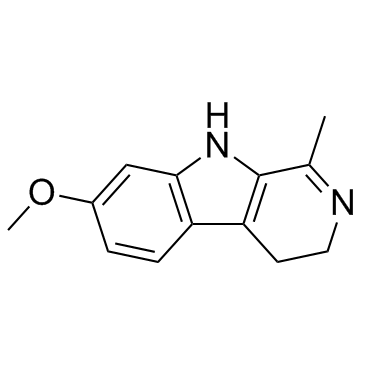
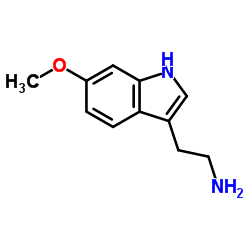
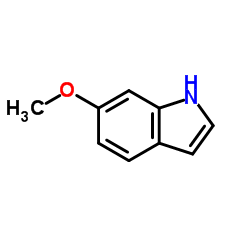
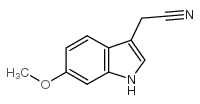
| Precursor 10 | |
|---|---|
| DownStream 10 | |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |



![1H-Pyrido[3,4-b]indole,2,3,4,9-tetrahydro-7-methoxy-1-methyl- (9CI) structure](https://image.chemsrc.com/caspic/429/486-93-1.png)






![9-benzyl-7-methoxy-1-methylpyrido[3,4-b]indole structure](https://image.chemsrc.com/caspic/198/17019-04-4.png)
![1-methyl-9H-pyrido[3,4-b]indol-7-ol hydrobromide structure](https://image.chemsrc.com/caspic/133/84625-56-9.png)
![1H-Pyrrolo[2,3-c]pyridine-2,3-dicarboxylicacid, 7-methyl structure](https://image.chemsrc.com/caspic/270/58795-15-6.png)