Betulin
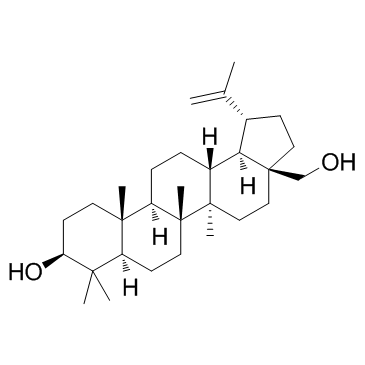
Betulin structure
|
Common Name | Betulin | ||
|---|---|---|---|---|
| CAS Number | 473-98-3 | Molecular Weight | 442.717 | |
| Density | 1.0±0.1 g/cm3 | Boiling Point | 522.3±23.0 °C at 760 mmHg | |
| Molecular Formula | C30H50O2 | Melting Point | 256-257 °C(lit.) | |
| MSDS | USA | Flash Point | 210.9±17.2 °C | |
| Symbol |

GHS08 |
Signal Word | Warning | |
|
A new cycloartane nortriterpenoid from stem and leaf of Quercus variabilis.
J. Asian Nat. Prod. Res. 15(9) , 1050-4, (2013) A new compound 3-acetyloxy-epicycloeucalenol-24-one (1), with 11 known compounds 3α-acetyloxy-4α,14α-dimethyl-9β,19-cycloergost-24-oic acid (2), 3-epicycloeucalenol (3), 3-epicycloeucalenyl-24-one (4), 3-epicycloeucalenyl acetate (5), 4β,14α-dimethyl-5α-ergos... |
|
|
Antidiabetic property of Aerva lanata (L.) Juss. ex Schult. is mediated by inhibition of alpha glucosidase, protein glycation and stimulation of adipogenesis.
J. Diabetes 7 , 548-61, (2015) Diabetes is the leading cause of morbidity and mortality, with a number currently diagnosed as high as 371 million. Plant-based therapy could be an ideal choice because of fewer side-effects and wider acceptability. Hence, the antihyperglycemic potential of A... |
|
|
alpha-Glucosidase inhibitory activity of triterpenoids from Cichorium intybus.
J. Nat. Prod. 71 , 910-3, (2008) Two new triterpenoids, 18alpha,19beta-20(30)-taraxasten-3beta,21alpha-diol (cichoridiol) (1) and 17-epi-methyl-6-hydroxyangolensate (intybusoloid) (2), were obtained from the methanolic extract of seeds of Cichorium intybus along with 11 known compounds, lupe... |
|
|
Chemical constituents of Caesalpinia decapetala (Roth) Alston.
Molecules 18(1) , 1325-36, (2013) The current study targets the chemical constituents of Caesalpinia decapetala (Roth) Alston and investigates the bioactivities of the isolated compounds. Fourteen known compounds were isolated using column chromatography, and structural identification was per... |
|
|
Efficient synthesis and biological evaluation of epiceanothic acid and related compounds.
Bioorg. Med. Chem. Lett. 21 , 338-41, (2011) Epiceanothic acid (1) is a naturally occurring, but very rare pentacyclic triterpene with a unique pentacyclic triterpene (PT) structure. An efficient synthesis of 1 starting from betulin (3) has been accomplished in 12-steps with a total yield of 10% in our ... |
|
|
Identification of pentacyclic triterpenes derivatives as potent inhibitors against glycogen phosphorylase based on 3D-QSAR studies.
Eur. J. Med. Chem. 46 , 2011-21, (2011) Naturally occurring pentacyclic triterpenes (PT), a novel class of inhibitors against glycogen phosphorylase (GP), hold promise for the treatment of type-2 diabetes and other diseases with disorders in glycogen metabolism. To identify novel and more potent GP... |
|
|
Study of skin anti-ageing and anti-inflammatory effects of dihydroquercetin, natural triterpenoids, and their synthetic derivatives.
Bioorg. Khim. 38(3) , 374-81, (2012) Accessible triterpenoids of ursane and lupane series, the flavonoid dihydroquercetin and their synthetic derivatives with polar substituentss were tested in vitro for inhibition of collagenase 1 (MMP-1) in UVB irradiation assay. Ursolic acid and uvaol disucci... |
|
|
Naturally occurring pentacyclic triterpenes as inhibitors of glycogen phosphorylase: synthesis, structure-activity relationships, and X-ray crystallographic studies.
J. Med. Chem. 51 , 3540-54, (2008) Twenty-five naturally occurring pentacyclic triterpenes, 15 of which were synthesized in this study, were biologically evaluated as inhibitors of rabbit muscle glycogen phosphorylase a (GPa). From SAR studies, the presence of a sugar moiety in triterpene sapo... |
|
|
Antiproliferative triterpenes from Melaleuca ericifolia.
J. Nat. Prod. 71 , 1787-90, (2008) Three new 28-norlupane triterpenes, 28-norlup-20(29)-en-3beta-hydroxy-17beta-hydroperoxide (1), 28-norlup-20(29)-en-3beta-hydroxy-17alpha-hydroperoxide (2), and 20 S-17beta,29-epoxy-28-norlupan-3beta-ol (3), were isolated from the leaves of Melaleuca ericifol... |
|
|
Structure-activity relationship study of betulinic acid, a novel and selective TGR5 agonist, and its synthetic derivatives: potential impact in diabetes.
J. Med. Chem. 53 , 178-190, (2010) We describe here the biological screening of a collection of natural occurring triterpenoids against the G protein-coupled receptor TGR5, known to be activated by bile acids and which mediates some important cell functions. This work revealed that betulinic (... |