| Structure | Name/CAS No. | Articles |
|---|---|---|
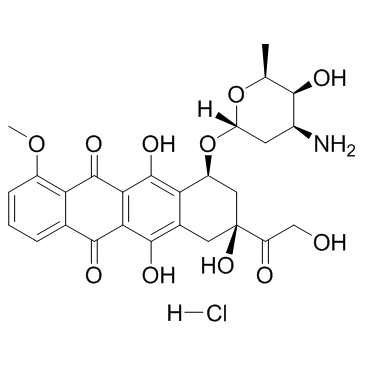 |
Doxorubicin Hydrochloride
CAS:25316-40-9 |
|
 |
Caffeine
CAS:58-08-2 |
|
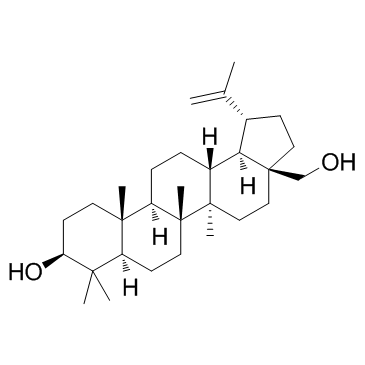 |
Betulin
CAS:473-98-3 |