TOAC
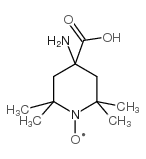
TOAC structure
|
Common Name | TOAC | ||
|---|---|---|---|---|
| CAS Number | 15871-57-5 | Molecular Weight | 215.26900 | |
| Density | 1.1255 (rough estimate) | Boiling Point | 355.55°C (rough estimate) | |
| Molecular Formula | C10H19N2O3 | Melting Point | 230 °C | |
| MSDS | Chinese USA | Flash Point | N/A | |
| Symbol |

GHS07 |
Signal Word | Warning | |
|
In silico interpretation of cw-ESR at 9 and 95 GHz of mono- and bis- TOAC-labeled Aib-homopeptides in fluid and frozen acetonitrile.
J. Phys. Chem. B 115(44) , 13026-36, (2011) In this paper, we address the interpretation of molecular properties of selected singly and doubly spin-labeled peptides from continuous-wave electron spin resonance (cw-ESR) spectroscopy. This study is performed by means of an integrated computational approa... |
|
|
Intramembrane water associated with TOAC spin-labeled alamethicin: electron spin-echo envelope modulation by D2O.
Biophys. J. 96(3) , 997-1007, (2009) Alamethicin is a 20-residue, hydrophobic, helical peptide, which forms voltage-sensitive ion channels in lipid membranes. The helicogenic, nitroxyl amino acid TOAC was substituted isosterically for Aib at residue positions 1, 8, or 16 in a F50/5 alamethicin a... |
|
|
Probing the helical tilt and dynamic properties of membrane-bound phospholamban in magnetically aligned bicelles using electron paramagnetic resonance spectroscopy.
Biochim. Biophys. Acta 1818(3) , 645-50, (2012) Wild-type phospholamban (WT-PLB), a Ca(2+)-ATPase (SERCA) regulator in the sarcoplasmic reticulum membrane, was studied using TOAC nitroxide spin labeling, magnetically aligned bicelles, and electron paramagnetic resonance (EPR) spectroscopy to ascertain stru... |
|
|
Membrane interaction of neuropeptide Y detected by EPR and NMR spectroscopy.
Biochim. Biophys. Acta 1714(2) , 103-13, (2005) Neuropeptide Y (NPY) is one of the most abundant peptides in the central nervous system of mammals. It belongs to the best-conserved peptides in nature, i.e., the amino acid sequences of even evolutionary widely separated species are very similar to each othe... |
|
|
Electron paramagnetic resonance studies of an integral membrane peptide inserted into aligned phospholipid bilayer nanotube arrays.
J. Am. Chem. Soc. 128(37) , 12070-1, (2006) This communication reports for the first time the determination of the helical tilt of an integral membrane peptide inserted into aligned phospholipids bilayer nanotube arrays using spin label EPR spectroscopy. Also, we demonstrate herein how the helical tilt... |
|
|
Synthesis and preliminary conformational analysis of TOAC spin-labeled analogues of the medium-length peptaibiotic tylopeptin B.
J. Pept. Sci. 18(1) , 37-44, (2012) A set of analogues of the 14-residue peptaibol tylopeptin B, containing the stable free-radical 4-amino-1-oxyl-2,2,6,6,-tetramethylpiperidine-4-carboxylic acid (TOAC) at one or two selected positions, was synthesized by the solid-phase methodology. A solution... |
|
|
Conformational properties of angiotensin II and its active and inactive TOAC-labeled analogs in the presence of micelles. Electron paramagnetic resonance, fluorescence, and circular dichroism studies.
Biopolymers 92(6) , 525-37, (2009) The interaction between angiotensin II (AII, DRVYIHPF) and its analogs carrying 2,2,6,6-tetramethylpiperidine-1-oxyl-4-amino-4-carboxylic acid (TOAC) and detergents--negatively charged sodium dodecyl sulfate (SDS) and zwitterionic N-hexadecyl-N,N-dimethyl-3-a... |
|
|
Distance measurements on a dual-labeled TOAC AChR M2δ peptide in mechanically aligned DMPC bilayers via dipolar broadening CW-EPR spectroscopy.
J. Phys. Chem. B 116(12) , 3866-73, (2012) A membrane alignment technique has been used to measure the distance between two TOAC nitroxide spin labels on the membrane-spanning M2δ, peptide of the nicotinic acetylcholine receptor (AChR), via CW-EPR spectroscopy. The TOAC-labeled M2δ peptides were mecha... |
|
|
Total syntheses in solution of TOAC-labelled alamethicin F50/5 analogues.
Chem. Biodivers. 4(6) , 1183-99, (2007) Total syntheses in solution of a set of four selected analogues of the 19-mer component F50/5 of alamethicin, the most extensively studied among the channel-former peptaibol antibiotics, are planned and reported. All analogues bear three Glu(OMe) residues, re... |
|
|
Conformational analysis of TOAC-labelled alamethicin F50/5 analogues.
Chem. Biodivers. 4(6) , 1256-68, (2007) In the preceding paper in this issue, we reported the total syntheses in solution of a set of four TOAC-containing analogues of the [L-Glu(OMe)(7,18,19)] F50/5 component of alamethicin, the prototype of peptaibol antibiotics forming channels in the biological... |