Synthesis and preliminary conformational analysis of TOAC spin-labeled analogues of the medium-length peptaibiotic tylopeptin B.
Marina Gobbo, Elisabetta Merli, Barbara Biondi, Simona Oancea, Antonio Toffoletti, Fernando Formaggio, Claudio Toniolo
Index: J. Pept. Sci. 18(1) , 37-44, (2012)
Full Text: HTML
Abstract
A set of analogues of the 14-residue peptaibol tylopeptin B, containing the stable free-radical 4-amino-1-oxyl-2,2,6,6,-tetramethylpiperidine-4-carboxylic acid (TOAC) at one or two selected positions, was synthesized by the solid-phase methodology. A solution conformational analysis performed by FTIR absorption and CD suggests that, in membrane-mimicking solvents, the labeled tylopeptin B analogues preserve the helical propensity of the parent peptide, with a preference for the α-helix or the 3(10) -helix type depending upon the nature of the solvent. In aqueous environment, the spin-labeled analogues present a higher content of helical conformation as a consequence of the strong helix promoter effect of the conformationally constrained TOAC residue. We observed a progressive increase of the quenching effect of the nitroxyl radical on the fluorescence of the N-terminal tryptophan as TOAC replaces the Aib residue at positions 13, 8, and 4, respectively. A membrane permeabilization assay performed on two selected analogues, TOAC(8) - and TOAC(13) -tylopeptin B, showed that the labeled peptides exhibit membrane-modifying properties comparable with those of the natural peptaibiotic. We conclude that our TOAC paramagnetic analogues of tylopeptin B are good models for a detailed ESR investigation of the mechanism of membrane permeabilization induced by medium-length peptaibiotics.Copyright © 2011 European Peptide Society and John Wiley & Sons, Ltd.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
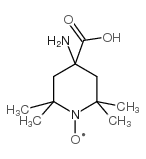 |
TOAC
CAS:15871-57-5 |
C10H19N2O3 |
|
In silico interpretation of cw-ESR at 9 and 95 GHz of mono- ...
2011-11-10 [J. Phys. Chem. B 115(44) , 13026-36, (2011)] |
|
Intramembrane water associated with TOAC spin-labeled alamet...
2009-02-01 [Biophys. J. 96(3) , 997-1007, (2009)] |
|
Probing the helical tilt and dynamic properties of membrane-...
2012-03-01 [Biochim. Biophys. Acta 1818(3) , 645-50, (2012)] |
|
Membrane interaction of neuropeptide Y detected by EPR and N...
2005-08-15 [Biochim. Biophys. Acta 1714(2) , 103-13, (2005)] |
|
Electron paramagnetic resonance studies of an integral membr...
2006-09-20 [J. Am. Chem. Soc. 128(37) , 12070-1, (2006)] |