2-氨基-3-(5-氟-1H-吲哚-3-基)丙酸
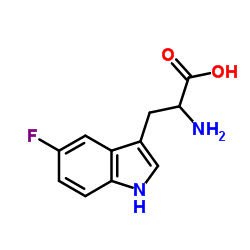
2-氨基-3-(5-氟-1H-吲哚-3-基)丙酸结构式

|
常用名 | 2-氨基-3-(5-氟-1H-吲哚-3-基)丙酸 | 英文名 | DL-5-Fluorotryptophan |
|---|---|---|---|---|
| CAS号 | 154-08-5 | 分子量 | 222.216 | |
| 密度 | 1.4±0.1 g/cm3 | 沸点 | 450.7±45.0 °C at 760 mmHg | |
| 分子式 | C11H11FN2O2 | 熔点 | 265 °C (dec.)(lit.) | |
| MSDS | 中文版 美版 | 闪点 | 226.4±28.7 °C |
|
Hydrophobic effect and hydrogen bonds account for the improved activity of a complement inhibitor, compstatin.
J. Med. Chem. 49(15) , 4616-22, (2006) Tryptophans at positions 4 and 7 of compstatin, a peptide complement inhibitor, are crucial for its interaction with C3. However, the nature of their involvement has not been studied to date. Here we investigate the molecular forces involved in the C3-compsta... |
|
|
Ionization potentials of fluoroindoles and the origin of nonexponential tryptophan fluorescence decay in proteins.
J. Am. Chem. Soc. 127(11) , 4104-13, (2005) This work reports an explanation for the unusual monoexponential fluorescence decay of 5-fluorotryptophan (5FTrp) in single-Trp mutant proteins [Broos, J.; Maddalena, F.; Hesp, B. H. J. Am. Chem. Soc. 2004, 126, 22-23] and substantially clarifies the origin o... |
|
|
In vivo synthesized proteins with monoexponential fluorescence decay kinetics.
J. Am. Chem. Soc. 126(1) , 22-3, (2004) Tryptophan, when in a protein, typically shows multiexponential fluorescence decay kinetics. Complex kinetics prevents a straightforward interpretation of time-resolved fluorescence protein data, particularly in anisotropy studies or if the effect of a dynami... |
|
|
5-fluoro-D,L-tryptophan as a dual NMR and fluorescent probe of α-synuclein.
Methods Mol. Biol. 895 , 197-209, (2012) Analysis of conventional proton nuclear magnetic resonance (NMR) experiments on intrinsically disordered proteins (IDPs) is challenging because of the highly flexible and multiple rapidly exchanging conformations typifying this class of proteins. One method t... |
|
|
[5-fluoro-tryptophan-containing N-terminal domain of the alpha-subunit of the Torpedo californica acetylcholine receptor: preparation in E. coli and 19F NMR study].
Bioorg. Khim. 29(4) , 384-90, (2003) A protein corresponding to the extracellular 1-209 domain of the alpha-subunit of the nicotine acetylcholine receptor from the electric organ of Torpedo californica was prepared using the corresponding cDNA domain by culturing Escherichia coli cells on a synt... |
|
|
Determination of the 19F NMR chemical shielding tensor and crystal structure of 5-fluoro-dl-tryptophan.
J. Magn. Reson. 187(1) , 88-96, (2007) 5-Fluoro-dl-tryptophan (5F-Trp) is a very sensitive probe used to investigate orientation and dynamics of biomacromolecules at the in situ level. In order to establish a (19)F NMR strategy, the crystal structure and (19)F chemical shielding tensor of 5F-Trp a... |
|
|
5-fluorotryptophan as dual probe for ground-state heterogeneity and excited-state dynamics in apoflavodoxin.
FEBS Lett. 583(17) , 2785-8, (2009) The apoflavodoxin protein from Azotobacter vinelandii harboring three tryptophan (Trp) residues, was biosynthetically labeled with 5-fluorotryptophan (5-FTrp). 5-FTrp has the advantage that chemical differences in its microenvironment can be sensitively visua... |
|
|
Molecular and biochemical studies of chondramide formation-highly cytotoxic natural products from Chondromyces crocatus Cm c5.
Chem. Biol. 13(6) , 667-81, (2006) The jaspamide/chondramide family of depsipeptides are mixed PKS/NRPS natural products isolated from marine sponges and a terrestrial myxobacterium that potently affect the function of the actin cytoskeleton. As a first step to improve production in heterologo... |
|
|
Sensitive high-performance liquid chromatographic assay for D-amino-acid oxidase activity in mammalian tissues using a fluorescent non-natural substrate, 5-fluoro-D-tryptophan.
J. Chromatogr. A. 1106(1-2) , 159-64, (2006) A sensitive assay for D-amino-acid oxidase (DAO) activity in mammalian tissues has been established. D-Tryptophan (D-Trp) analogs were tested as substrates for DAO, and 5-fluoro-D-tryptophan (D-FTP) was found to be the best substrate. By the enzymatic reactio... |
|
|
Structural investigation of the transmembrane C domain of the mannitol permease from Escherichia coli using 5-FTrp fluorescence spectroscopy.
Biochim. Biophys. Acta 1818(3) , 861-8, (2012) The mannitol transporter EII(mtl) from Escherichia coli is responsible for the uptake of mannitol over the inner membrane and its concomitant phosphorylation. EII(mtl) is functional as a dimer and its membrane-embedded C domain, IIC(mtl), harbors one high aff... |