| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
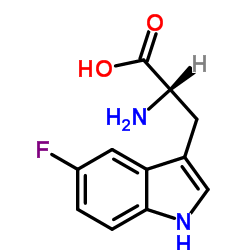 |
(S)-2-氨基-3-(5-氟-1H-吲哚-3-基)丙酸
CAS:16626-02-1 |
|
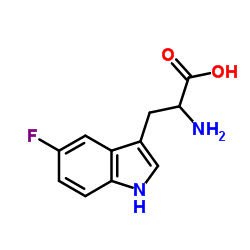 |
2-氨基-3-(5-氟-1H-吲哚-3-基)丙酸
CAS:154-08-5 |