coniferyl aldehyde
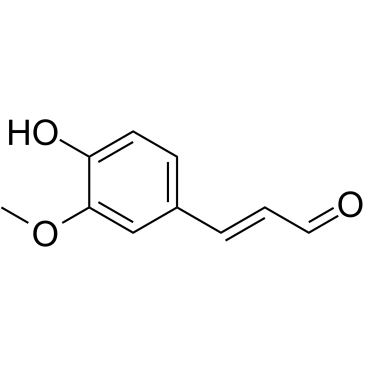
coniferyl aldehyde structure
|
Common Name | coniferyl aldehyde | ||
|---|---|---|---|---|
| CAS Number | 458-36-6 | Molecular Weight | 178.185 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 338.8±27.0 °C at 760 mmHg | |
| Molecular Formula | C10H10O3 | Melting Point | 80-82ºC(lit.) | |
| MSDS | Chinese USA | Flash Point | 136.8±17.2 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of coniferyl aldehydeConiferaldehyde (Ferulaldehyde), isolated from the ethanol extract of Vitex rotundifolia fruits, is an effective inducer of heme oxygenase-1 (HO-1). Coniferaldehyde exerts anti-inflammatory properties in response to LPS. Coniferaldehyde inhibits LPS-induced apoptosis through the PKCα/β II/Nrf-2/HO-1 dependent pathway in RAW264.7 macrophage cells[1]. |
| Name | coniferyl aldehyde |
|---|---|
| Synonym | More Synonyms |
| Description | Coniferaldehyde (Ferulaldehyde), isolated from the ethanol extract of Vitex rotundifolia fruits, is an effective inducer of heme oxygenase-1 (HO-1). Coniferaldehyde exerts anti-inflammatory properties in response to LPS. Coniferaldehyde inhibits LPS-induced apoptosis through the PKCα/β II/Nrf-2/HO-1 dependent pathway in RAW264.7 macrophage cells[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 338.8±27.0 °C at 760 mmHg |
| Melting Point | 80-82ºC(lit.) |
| Molecular Formula | C10H10O3 |
| Molecular Weight | 178.185 |
| Flash Point | 136.8±17.2 °C |
| Exact Mass | 178.062988 |
| PSA | 46.53000 |
| LogP | 1.35 |
| Vapour Pressure | 0.0±0.8 mmHg at 25°C |
| Index of Refraction | 1.593 |
| Storage condition | 2-8°C |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi: Irritant; |
| Risk Phrases | 36/37/38 |
| Safety Phrases | 26-37/39 |
| RIDADR | NONH for all modes of transport |
| HS Code | 2912499000 |
| Precursor 7 | |
|---|---|
| DownStream 10 | |
| HS Code | 2912499000 |
|---|---|
| Summary | 2912499000. other aldehyde-ethers, aldehyde-phenols and aldehydes with other oxygen function. VAT:17.0%. Tax rebate rate:9.0%. . MFN tariff:5.5%. General tariff:30.0% |
|
A derivatization and validation strategy for determining the spatial localization of endogenous amine metabolites in tissues using MALDI imaging mass spectrometry.
J. Mass Spectrom. 49(8) , 665-73, (2014) Imaging mass spectrometry (IMS) studies increasingly focus on endogenous small molecular weight metabolites and consequently bring special analytical challenges. Since analytical tissue blanks do not ... |
|
|
Reagent precoated targets for rapid in-tissue derivatization of the anti-tuberculosis drug isoniazid followed by MALDI imaging mass spectrometry.
J. Am. Soc. Mass Spectrom. 22(8) , 1409-19, (2011) Isoniazid (INH) is an important component of front-line anti-tuberculosis therapy with good serum pharmacokinetics but unknown ability to penetrate tuberculous lesions. However, endogenous background ... |
|
|
Antifungal synergistic effect of scopoletin, a hydroxycoumarin isolated from Melia azedarach L. fruits.
J. Agric. Food Chem. 53(8) , 2922-7, (2005) In the continuous search for antifungal compounds from plants, the hydroxycoumarin scopoletin (1) was isolated from seed kernels of Melia azedarach L. from which three other compounds, vanillin (2), 4... |
| Ferulaldehyde |
| (2E)-3-(4-hydroxy-3-methoxyphenyl)prop-2-enal |
| (E)-3-(4-hydroxy-3-methoxyphenyl)prop-2-enal |
| Ferulyl aldehyde |
| 2-Propenal, 3-(4-hydroxy-3-methoxyphenyl), (E)- |
| 4-hydroxy-3-methoxy-benzaldehyde |
| MFCD00075811 |
| 4-hydroxy-3-methoxy-cinnamon-aldehyde |
| 2-Propenal, 3-(4-hydroxy-3-methoxyphenyl)-, (2E)- |
| coniferaldehyde |
| 4-hydroxy-3-methoxycinnamic aldehyde |
| EINECS 207-278-4 |
| 4-HYDROXY-3-METHOXYCINNAMALDEHYDE |
| (2E)-3-(4-Hydroxy-3-methoxyphenyl)acrylaldehyde |
| coniferyl aldehyde |
| Coniferylaldehyde |
 CAS#:458-35-5
CAS#:458-35-5 CAS#:65401-83-4
CAS#:65401-83-4 CAS#:97-53-0
CAS#:97-53-0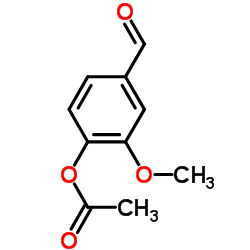 CAS#:881-68-5
CAS#:881-68-5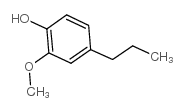 CAS#:2785-87-7
CAS#:2785-87-7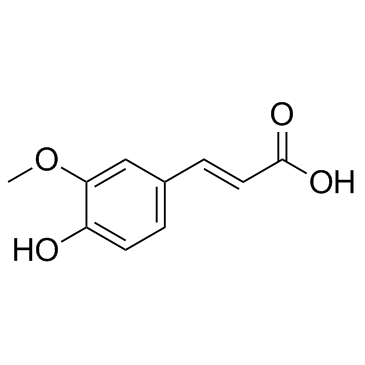 CAS#:1135-24-6
CAS#:1135-24-6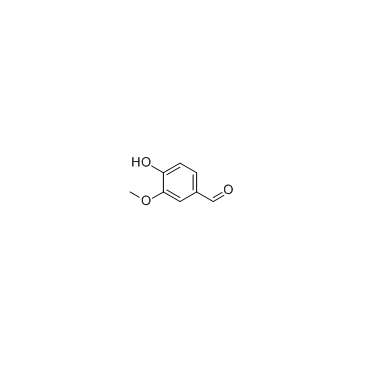 CAS#:121-33-5
CAS#:121-33-5 CAS#:2305-13-7
CAS#:2305-13-7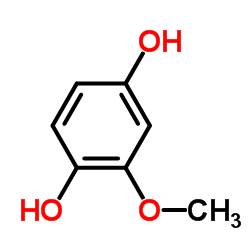 CAS#:824-46-4
CAS#:824-46-4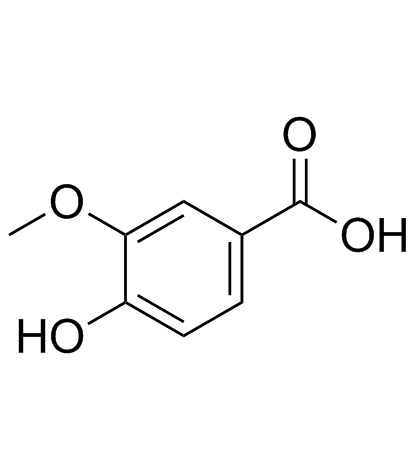 CAS#:121-34-6
CAS#:121-34-6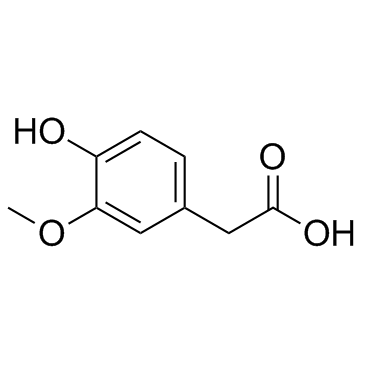 CAS#:306-08-1
CAS#:306-08-1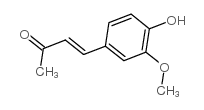 CAS#:1080-12-2
CAS#:1080-12-2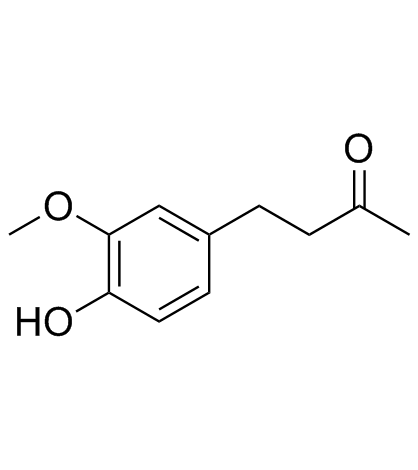 CAS#:122-48-5
CAS#:122-48-5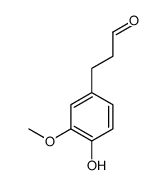 CAS#:80638-48-8
CAS#:80638-48-8 CAS#:78954-23-1
CAS#:78954-23-1
