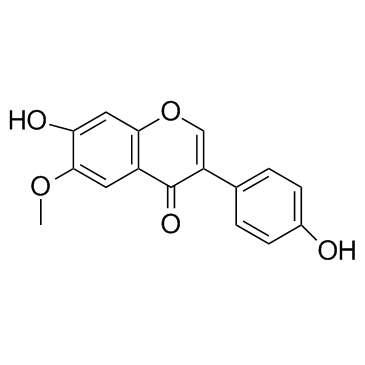
Glycitin

Glycitin structure
|
Common Name | Glycitin | ||
|---|---|---|---|---|
| CAS Number | 40246-10-4 | Molecular Weight | 446.404 | |
| Density | 1.5±0.1 g/cm3 | Boiling Point | 751.1±60.0 °C at 760 mmHg | |
| Molecular Formula | C22H22O10 | Melting Point | 210ºC | |
| MSDS | Chinese USA | Flash Point | 264.1±26.4 °C | |
Use of GlycitinGlycitin is a natural isoflavone isolated from legumes; promotes the proliferation of bone marrow stromal cells and osteoblasts and suppresses bone turnover. |
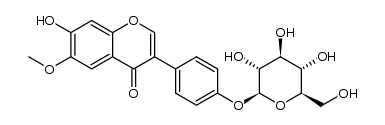
| Name | 3-(4-hydroxyphenyl)-6-methoxy-7-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxychromen-4-one |
|---|---|
| Synonym | More Synonyms |
| Description | Glycitin is a natural isoflavone isolated from legumes; promotes the proliferation of bone marrow stromal cells and osteoblasts and suppresses bone turnover. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.5±0.1 g/cm3 |
|---|---|
| Boiling Point | 751.1±60.0 °C at 760 mmHg |
| Melting Point | 210ºC |
| Molecular Formula | C22H22O10 |
| Molecular Weight | 446.404 |
| Flash Point | 264.1±26.4 °C |
| Exact Mass | 446.121307 |
| PSA | 159.05000 |
| LogP | 0.16 |
| Vapour Pressure | 0.0±2.6 mmHg at 25°C |
| Index of Refraction | 1.675 |
| Storage condition | Store at -20 |
|
~6% 
Glycitin CAS#:40246-10-4 |
| Literature: Shimoda, Kei; Hamada, Hiroki Molecules, 2010 , vol. 15, # 8 p. 5153 - 5161 |
|
~% 
Glycitin CAS#:40246-10-4 |
| Literature: Nogradi, Mihaly; Szoellosy, Aron Liebigs Annales, 1996 , # 10 p. 1651 - 1652 |
|
~68% 
Glycitin CAS#:40246-10-4 |
| Literature: Nogradi, Mihaly; Szoellosy, Aron Liebigs Annales, 1996 , # 10 p. 1651 - 1652 |
|
~% 
Glycitin CAS#:40246-10-4 |
| Literature: Nogradi, Mihaly; Szoellosy, Aron Liebigs Annales, 1996 , # 10 p. 1651 - 1652 |
|
~% 
Glycitin CAS#:40246-10-4 |
| Literature: Nogradi, Mihaly; Szoellosy, Aron Liebigs Annales, 1996 , # 10 p. 1651 - 1652 |
|
~94% 
Glycitin CAS#:40246-10-4 |
| Literature: Al-Maharik, Nawaf; Botting, Nigel P. European Journal of Organic Chemistry, 2008 , # 33 p. 5622 - 5629 |
|
~% 
Glycitin CAS#:40246-10-4 |
| Literature: Nogradi, Mihaly; Szoellosy, Aron Liebigs Annales, 1996 , # 10 p. 1651 - 1652 |
|
Effects of naturally occurring isoflavones on prostaglandin E2 production.
Planta Med. 68 , 97-100, (2002) Previously, we reported that the isoflavones tectorigenin and tectoridin, a glycosylated tectorigenin, isolated from the rhizomes of Belamcanda chinensis have an activity to inhibit prostaglandin (PG)... |
|
|
Isoflavone glycosides from the flowers of Pueraria thunbergiana. Park HJ
Phytochemistry 51 , 147-151, (1999)
|
| BIDD:ER0468 |
| 4',7-Dihydroxy-6-methoxyisoflavone-7-D-glucoside |
| 3-(4-Hydroxyphenyl)-6-methoxy-4-oxo-4H-chromen-7-yl b-D-glucopyranoside |
| MFCD00800711 |
| Glycitein 7-O-|A-glucoside |
| Glycitein-7-|A-O-glucoside |
| Glycitein 7-O-β-D-glucoside |
| 3-(4-Hydroxyphenyl)-6-methoxy-4-oxo-4H-chromen-7-yl β-D-glucopyranoside |
| 4H-1-Benzopyran-4-one |
| 4H-1-Benzopyran-4-one, 7-(β-D-glucopyranosyloxy)-3-(4-hydroxyphenyl)-6-methoxy- |
| Glycitin |
| Glycitein 7-O-glucoside |
![1-[5-Methoxy-2,4-bis(phenylmethoxy)phenyl]-ethanone structure](https://image.chemsrc.com/caspic/390/7298-22-8.png)


![3,3-Dimethoxy-1-[5-Methoxy-1,4-bis(phenylmethoxy)phenyl]-2-[4-(phenylmethoxy)phenyl]-1-propanone structure](https://image.chemsrc.com/caspic/342/58115-19-8.png)

