Hyoscyamine
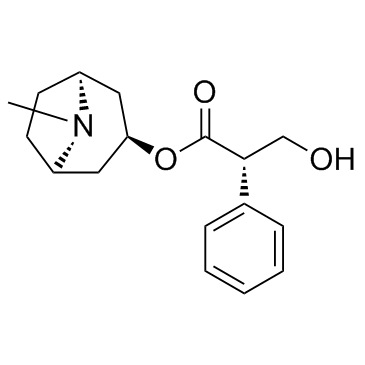
Hyoscyamine structure
|
Common Name | Hyoscyamine | ||
|---|---|---|---|---|
| CAS Number | 101-31-5 | Molecular Weight | 289.369 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 429.8±45.0 °C at 760 mmHg | |
| Molecular Formula | C17H23NO3 | Melting Point | 108.5ºC | |
| MSDS | N/A | Flash Point | 213.7±28.7 °C | |
Use of HyoscyamineL-Hyoscyamine is a chemical compound, a tropane alkaloid it is the levo-isomer to atropine.Target: mAChRHyoscyamine is a chemical compound, a tropane alkaloid it is the levo-isomer to atropine. It is a secondary metabolite of some plants, particularly henbane (Hyoscamus niger.)Hyoscyamine is used to provide symptomatic relief to various gastrointestinal disorders including spasms, peptic ulcers, irritable bowel syndrome, pancreatitis, colic and cystitis. It has also been used to relieve some heart problems, control some of the symptoms of Parkinson's disease, as well as for control of respiratory secretions in end of life care [1]. |
| Name | (S)-atropine |
|---|---|
| Synonym | More Synonyms |
| Description | L-Hyoscyamine is a chemical compound, a tropane alkaloid it is the levo-isomer to atropine.Target: mAChRHyoscyamine is a chemical compound, a tropane alkaloid it is the levo-isomer to atropine. It is a secondary metabolite of some plants, particularly henbane (Hyoscamus niger.)Hyoscyamine is used to provide symptomatic relief to various gastrointestinal disorders including spasms, peptic ulcers, irritable bowel syndrome, pancreatitis, colic and cystitis. It has also been used to relieve some heart problems, control some of the symptoms of Parkinson's disease, as well as for control of respiratory secretions in end of life care [1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 429.8±45.0 °C at 760 mmHg |
| Melting Point | 108.5ºC |
| Molecular Formula | C17H23NO3 |
| Molecular Weight | 289.369 |
| Flash Point | 213.7±28.7 °C |
| Exact Mass | 289.167786 |
| PSA | 49.77000 |
| LogP | 1.53 |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.581 |
| Storage condition | 2-8°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Hazard Codes | T+: Very toxic; |
|---|---|
| Risk Phrases | R26/28 |
| Safety Phrases | 24-45 |
| RIDADR | UN 1544 6 |
| WGK Germany | 3 |
| RTECS | NH0875000 |
| HS Code | 2933990090 |
|
~% 
Hyoscyamine CAS#:101-31-5 |
| Literature: Phytochemistry (Elsevier), , vol. 28, # 12 p. 3507 - 3510 |
|
~20% 
Hyoscyamine CAS#:101-31-5 |
| Literature: Journal of Natural Products, , vol. 69, # 9 p. 1335 - 1340 |
|
~% 
Hyoscyamine CAS#:101-31-5 |
| Literature: Archiv der Pharmazie (Weinheim, Germany), , vol. 240, p. 501 |
|
~% 
Hyoscyamine CAS#:101-31-5 |
| Literature: Journal of the Chemical Society, , vol. 101, p. 950 |
|
~% 
Hyoscyamine CAS#:101-31-5 |
| Literature: Journal of the Chemical Society, , vol. 95, p. 1966 |
|
~% 
Hyoscyamine CAS#:101-31-5 |
| Literature: Chemistry Letters, , p. 983 - 986 |
| Precursor 7 | |
|---|---|
| DownStream 5 | |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Role of 17-beta estradiol in baroreflex sensitivity in the nucleus tractus solitarii via the autonomic system in ovariectomized rats.
Neurosciences (Riyadh.) 18(2) , 126-32, (2013) To investigate the effect of estrogen exerted through the autonomic system in the nucleus tractus solitarii (NTS) on increasing the sensitivity of the baroreflex under conditions of acute hypertension... |
|
|
Comparison of subcellular distribution and functions between exogenous and endogenous M1 muscarinic acetylcholine receptors.
Life Sci. 93(1) , 17-23, (2013) Recombinant systems have been used for evaluating the properties of G-protein-coupled receptors (GPCRs) on the assumption of cell surface expression. However, many GPCRs, including muscarinic acetylch... |
|
|
Sudden unexpected death in a mouse model of Dravet syndrome.
J. Clin. Invest. 123(4) , 1798-808, (2013) Sudden unexpected death in epilepsy (SUDEP) is the most common cause of death in intractable epilepsies, but physiological mechanisms that lead to SUDEP are unknown. Dravet syndrome (DS) is an infanti... |
| CYSTOSPAZ |
| L-Hyoscyamine |
| 3a-Tropanyl S-(-)-Tropate |
| (3-endo)-8-Methyl-8-azabicyclo[3.2.1]oct-3-yl-(2S)-3-hydroxy-2-phenylpropanoat |
| scopolamine HCl |
| SCOPINE TROPATE |
| Levsin |
| (3-endo)-8-Methyl-8-azabicyclo[3.2.1]oct-3-yl (2S)-3-hydroxy-2-phenylpropanoate |
| (S)-(-)-hyoscyamine |
| Benzeneacetic acid, α-(hydroxymethyl)-, (3-endo)-8-methyl-8-azabicyclo[3.2.1]oct-3-yl ester, (αS)- |
| SCOPINE TROPATE HYDROCHLORIDE |
| Benzeneacetic acid, α-(hydroxymethyl)-, (3-endo)-8-methyl-8-azabicyclo(3.2.1)oct-3-yl ester, (αS)- |
| MFCD00067306 |
| (S)-atropine |
| Hyoscyamine |
| (-)-Hycosamine |
| (-)-Hyoscyamine |
| EINECS 202-933-0 |
| Daturine |
| chlorhydratedescopolamine |
| Egacene |
| 1-Tropic Acid Ester with Tropine |
| (-)-atropine |
| er),hcl |
| [3(S)-endo]-a-(Hydroxymethyl)benzeneacetic Acid 8-Methyl-8-azabicyclo[3.2.1]oct-3-yl Ester |
| Peptard |
| ScopolamineSeries |
| L-Tropine tropate |
| (2S)-(1R,5S)-8-Methyl-8-azabicyclo[3.2.1]octan-3-yl 3-hydroxy-2-phenylpropanoate |
| 1aH,5aH-Tropan-3a-ol(-)-tropate (Ester) |
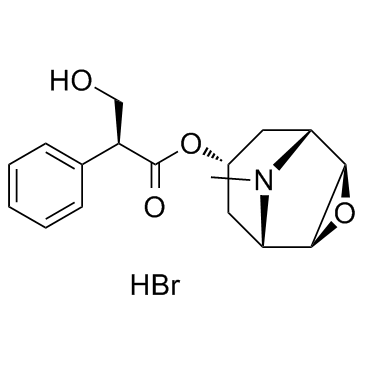

 CAS#:529-64-6
CAS#:529-64-6 CAS#:5627-14-5
CAS#:5627-14-5![8-Methyl-8-azabicyclo[3.2.1]oct-3-yl atropate structure](https://image.chemsrc.com/caspic/463/500-55-0.png) CAS#:500-55-0
CAS#:500-55-0
