Lomustine
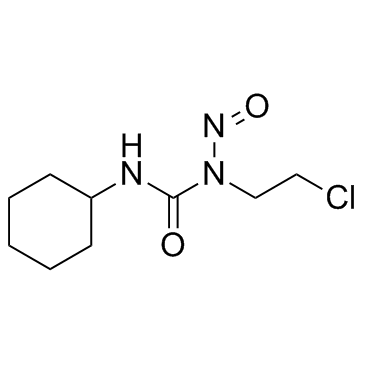
Lomustine structure
|
Common Name | Lomustine | ||
|---|---|---|---|---|
| CAS Number | 13010-47-4 | Molecular Weight | 233.695 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | N/A | |
| Molecular Formula | C9H16ClN3O2 | Melting Point | 88-90 | |
| MSDS | Chinese USA | Flash Point | N/A | |
| Symbol |


GHS06, GHS08 |
Signal Word | Danger | |
|
Expression of O(6)-methylguanine-DNA methyltransferase causes lomustine resistance in canine lymphoma cells.
Can. J. Vet. Res. 79 , 201-9, (2015) The DNA repair protein O (6)-methylguanine-DNA methyltransferase (MGMT) causes resistance to nitrosoureas in various human cancers. In this study, we analyzed the correlation between canine lymphomas and MGMT in vitro. Two of five canine lymphoma cell lines r... |
|
|
Preliminary study of lomustine in the treatment of intracranial masses in dogs following localization by imaging techniques.
Semin. Vet. Med. Surg. Small Anim. 5(4) , 241-5, (1990)
|
|
|
The nitrosoureas: carmustine (BCNU) and lomustine (CCNU).
Cancer Treat. Rev. 9(4) , 313-30, (1982)
|
|
|
Treatment of advanced neuroendocrine tumours using combination chemotherapy with lomustine and 5-fluorouracil.
Clin. Endocrinol. (Oxf.) 57(2) , 169-83, (2002) Combination chemotherapy with the two agents streptozotocin (SZT), which is a nitrosurea, and 5-fluorouracil (5-FU), an alkylating agent, has a long-established role in the treatment of neuroendocrine tumours; however, it is often accompanied by considerable ... |
|
|
The role of an all-oral chemotherapy containing lomustine (CCNU) in advanced,fs progressive Hodgkin lymphoma: a patient-friendly palliative option which can result in long-term disease control.
Ann. Oncol. 21(2) , 426-8, (2010)
|
|
|
High-dose etoposide in allogeneic stem cell transplantation.
Cancer Chemother. Pharmacol. 70(6) , 765-82, (2012) The anti-leukemic effect of etoposide is well documented. High-dose etoposide 60 mg/kg in combination with fractionated total body irradiation (TBI), usually single fractions of 1.2 Gy up to a total of 13.2 Gy, is used as conditioning therapy for allogeneic s... |
|
|
Bevacizumab alone at 5 mg/kg in an every-3-week schedule for patients with recurrent glioblastomas: a single center experience.
Tumori 99(5) , 601-3, (2013) The use of bevacizumab is increasingly reported in neuro-oncology. The most common schedule is 10 mg/kg every 2 weeks. We retrospectively investigated the efficacy of a 3-week schedule of 5 mg/kg bevacizumab in patients with recurrent glioblastomas. Fourteen ... |
|
|
Phase III trial of chemoradiotherapy for anaplastic oligodendroglioma: long-term results of RTOG 9402.
J. Clin. Oncol. 31(3) , 337-43, (2013) Anaplastic oligodendrogliomas, pure (AO) and mixed (anaplastic oligoastrocytoma [AOA]), are chemosensitive, especially if codeleted for 1p/19q, but whether patients live longer after chemoradiotherapy is unknown.Eligible patients with AO/AOA were randomly ass... |
|
|
Adjuvant procarbazine, lomustine, and vincristine chemotherapy in newly diagnosed anaplastic oligodendroglioma: long-term follow-up of EORTC brain tumor group study 26951.
J. Clin. Oncol. 31(3) , 344-50, (2013) Anaplastic oligodendroglioma are chemotherapy-sensitive tumors. We now present the long-term follow-up findings of a randomized phase III study on the addition of six cycles of procarbazine, lomustine, and vincristine (PCV) chemotherapy to radiotherapy (RT).A... |
|
|
Intrinsic molecular subtypes of glioma are prognostic and predict benefit from adjuvant procarbazine, lomustine, and vincristine chemotherapy in combination with other prognostic factors in anaplastic oligodendroglial brain tumors: a report from EORTC study 26951.
J. Clin. Oncol. 31(3) , 328-36, (2013) Intrinsic glioma subtypes (IGSs) are molecularly similar tumors that can be identified based on unsupervised gene expression analysis. Here, we have evaluated the clinical relevance of these subtypes within European Organisation for Research and Treatment of ... |