Phenethyl isocyanate
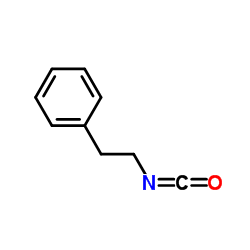
Phenethyl isocyanate structure
|
Common Name | Phenethyl isocyanate | ||
|---|---|---|---|---|
| CAS Number | 1943-82-4 | Molecular Weight | 147.174 | |
| Density | 1.0±0.1 g/cm3 | Boiling Point | 228.5±19.0 °C at 760 mmHg | |
| Molecular Formula | C9H9NO | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 67.4±22.6 °C | |
| Symbol |




GHS05, GHS06, GHS08, GHS09 |
Signal Word | Danger | |
|
High-performance liquid chromatographic resolution of enantiomers of 1-phenyl-2-aminopropanes (amphetamines) with four chiral reagents.
J. Chromatogr. A. 307 , 335, (1984) High-performance liquid chromatography (HPLC) was employed for resolution of enantiomers of chiral ring-substituted 1-phenyl-2- aminopropanes (amphetamines) and 1-phenylethylamine following derivatization with four chiral reagents: (R)-(+)-1-phenylethyl isocy... |
|
|
[The separation of some racemic beta-blockers and alpha-sympathomimetics with HPLC following derivatization].
Pharmazie 46(6) , 432-4, (1991) Five beta-adrenergic blocking agents and two alpha-sympathicomimetics were separated after derivatization with (R)-(-)-1-(Naphth-1-yl)-ethylisocyanate (NEIC). For two of these analytes the separation was successful also after reaction with (R)-(+)-1-phenyleth... |
|
|
A new synthesis of isocyanates. Gittos MW, et al.
J. Chem. Soc. Perkin Trans. I 2 , 141-3, (1976)
|
|
|
Role of cytochrome P4502E1-dependent formation of hydroxyethyl free radical in the development of liver damage in rats intragastrically fed with ethanol.
Hepatology 23(1) , 155-63, (1996) We have previously shown that the treatment with diallyl sulfide (DAS) and phenylethyl isothiocyanate (PIC) of rats receiving ethanol in the alcohol tube-feeding model effectively suppressed the induction of cytochrome P4502E1 (CYP2E1) by ethanol. Here we rep... |
|
|
Hydrogen sulfide protects colon cancer cells from chemopreventative agent beta-phenylethyl isothiocyanate induced apoptosis.
World J. Gastroenterol. 11(26) , 3990-7, (2005) Hydrogen sulfide (H(2)S) is a prominent gaseous constituent of the gastrointestinal (GI) tract with known cytotoxic properties. Endogenous concentrations of H(2)S are reported to range between 0.2-3.4 mmol/L in the GI tract of mice and humans. Considering suc... |
|
|
Silica gel high-performance liquid chromatography for the simultaneous determination of propranolol and 4-hydroxypropranolol enantiomers after chiral derivatization.
J. Chromatogr. A. 310(2) , 424-30, (1984)
|
|
|
Inhibition of human leukaemia 60 cell growth by mercapturic acid metabolites of phenylethyl isothiocyanate.
Food Chem. Toxicol. 34(4) , 385-92, (1996) Mercapturic acid pathway metabolites of phenylethyl isothiocyanate inhibited the growth of human leukaemia 60 (HL60) cells in vitro. The adduct with L-cysteine, S-(N-phenylethylthiocarbamoyl)cysteine, was the most potent with strong antileukaemic activity: th... |
|
|
Simultaneous determination of propranolol and 4-hydroxypropranolol enantiomers after chiral derivatization using reversed-phase high-performance liquid chromatography.
J. Chromatogr. A. 527(2) , 351-9, (1990) A reversed-phase high-performance liquid chromatographic method is described, which allows the simultaneous quantification of propranolol and 4-hydroxypropranolol enantiomers in human plasma. After extraction from plasma (pH 10.5) using ethyl acetate, the ena... |
|
|
Gas-chromatographic resolution of enantiomeric secondary alcohols. Stereoselective reductive metabolism of ketones in rabbit-liver cytosol.
Drug Metab. Dispos. 9(6) , 557-60, (1981) Chiral secondary alcohols were treated with (S)-(-)-1-phenylethyl isocyanate. For each racemic alcohol, the resulting diastereomeric urethane derivatives were resolved on flexible fused-silica capillary GLC columns with retention times of 15 min or less. Deri... |
|
|
1-phenylethyl isocyanate is a powerful reagent for the chiral analysis of secondary alcohols and hydroxy fatty acids with remote stereogenic centres.
J. Chromatogr. A. 1165(1-2) , 182-90, (2007) 1-phenylethyl isocyanate (1-PEIC), a chiral derivatisation reagent for the resolution of secondary alcohols is a powerful tool to determine the configuration and enantiomeric excess of medium- to long-chain secondary alcohols by capillary gas chromatography. ... |