Simultaneous determination of propranolol and 4-hydroxypropranolol enantiomers after chiral derivatization using reversed-phase high-performance liquid chromatography.
H G Schaefer, H Spahn, L M Lopez, H Derendorf
Index: J. Chromatogr. A. 527(2) , 351-9, (1990)
Full Text: HTML
Abstract
A reversed-phase high-performance liquid chromatographic method is described, which allows the simultaneous quantification of propranolol and 4-hydroxypropranolol enantiomers in human plasma. After extraction from plasma (pH 10.5) using ethyl acetate, the enantiomers are derivatized with R-(+)-phenylethylisocyanate as chiral derivatization reagent and triethylamine as basic catalyst in chloroform. Ascorbic acid is used to prevent 4-hydroxypropranolol from oxidation during the extraction. Chromatographic separation on ODS columns and fluorescence detection (228 nm/greater than 340 nm) allows sensitive quantitation of all derivatives. Incubation of the plasma samples with beta-glucuronidase/arylsulfatase and the use of the specific beta-glucuronidase inhibitor saccharo-1,4-lactone allows the quantitation of both the sulfate and glucuronide conjugates of the enantiomers. The method was applied to human plasma samples from a subject after administration of 60 mg racemic propranolol three times daily.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
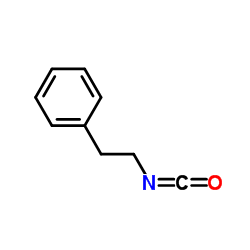 |
Phenethyl isocyanate
CAS:1943-82-4 |
C9H9NO |
|
High-performance liquid chromatographic resolution of enanti...
[J. Chromatogr. A. 307 , 335, (1984)] |
|
[The separation of some racemic beta-blockers and alpha-symp...
1991-06-01 [Pharmazie 46(6) , 432-4, (1991)] |
|
A new synthesis of isocyanates. Gittos MW, et al.
[J. Chem. Soc. Perkin Trans. I 2 , 141-3, (1976)] |
|
Role of cytochrome P4502E1-dependent formation of hydroxyeth...
1996-01-01 [Hepatology 23(1) , 155-63, (1996)] |
|
Hydrogen sulfide protects colon cancer cells from chemopreve...
2005-07-14 [World J. Gastroenterol. 11(26) , 3990-7, (2005)] |