Tocainide hydrochloride
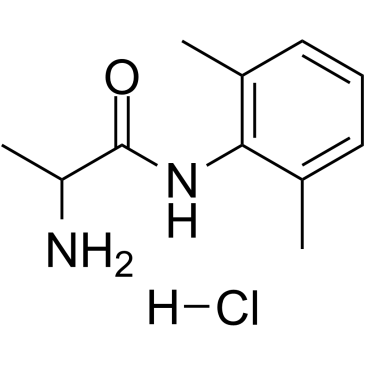
Tocainide hydrochloride structure
|
Common Name | Tocainide hydrochloride | ||
|---|---|---|---|---|
| CAS Number | 71395-14-7 | Molecular Weight | 192.25800 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C11H17ClN2O | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | N/A | |
| Symbol |

GHS07 |
Signal Word | Warning | |
|
Synthesis, structure and pharmacology of acyl-2,6-xylidines.
Acta Pol. Pharm. 61(3) , 215-21, (2004) L-2-perhydroheterocyclicalkyl acids were condensed with 2,6-xylidine. 8 new optically active acyl-2,6-xylidines were obtained. Absolute configuration of acyl-2,6-xylidines were selected for pharmacological examinations. |
|
|
Quantitative structure-pharmacokinetic relationships for drug clearance by using statistical learning methods.
J. Mol. Graph. Model. 24(5) , 383-95, (2006) Quantitative structure-pharmacokinetic relationships (QSPkR) have increasingly been used for the prediction of the pharmacokinetic properties of drug leads. Several QSPkR models have been developed to predict the total clearance (CL(tot)) of a compound. These... |
|
|
The efficacy of anticonvulsants on orofacial pain: a systematic review.
Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 111(5) , 627-33, (2011) Controversy exists about the effectiveness of anticonvulsants for the management of orofacial pain disorders. To ascertain appropriate therapies, a systematic review was conducted of existing randomized controlled trials.Trials were identified from PubMed, Co... |
|
|
Non-antiepileptic drugs for trigeminal neuralgia.
Cochrane Database Syst. Rev. (1) , CD004029, (2011) Non-antiepileptic drugs have been used in the management of trigeminal neuralgia since the 1970s.The objective was to systematically review the efficacy and tolerability of non-antiepileptic drugs for trigeminal neuralgia.For this updated review we searched t... |
|
|
Tocainide analogues binding to human serum albumin: a HPLAC and circular dichroism study.
J. Pharm. Biomed. Anal. 53(2) , 179-85, (2010) A series of synthesised tocainide analogues were characterized for their human serum albumin (HSA) binding, using high-performance liquid affinity chromatography (HPLAC) and circular dichroism (CD). The synthesis and physico-chemical characterization of compo... |
|
|
Effect of acidity on the enantiomeric resolution of thyroxine and tocainide by HPLC on a (+)-(18-crown-6)-2,3,11,12-tetracarboxylic acid column.
J. Biochem. Biophys. Methods 54(1-3) , 407-13, (2002) Enantiomeric resolution of thyroxine and tocainide was achieved on a (+)-(18-crown-6)-2,3,11,12-tetracarboxylic acid column. The mobile phases were methanol/water (4:1, v/v) and methanol/water containing 5 mM sulfuric acid (4:1, v/v) for tocainide and thyroxi... |
|
|
Increased rigidity of the chiral centre of tocainide favours stereoselectivity and use-dependent block of skeletal muscle Na(+) channels enhancing the antimyotonic activity in vivo.
Br. J. Pharmacol. 134(7) , 1523-31, (2001) 1. Searching for the structural requirements improving the potency and the stereoselectivity of Na(+) channel blockers as antimyotonic agents, new derivatives of tocainide, in which the chiral carbon atom is constrained in a rigid alpha-proline or pyrrolo-imi... |
|
|
Constrained analogues of tocainide as potent skeletal muscle sodium channel blockers towards the development of antimyotonic agents.
Eur. J. Med. Chem. 43(11) , 2535-40, (2008) 1-Benzyl-N-(2,6-dimethylphenyl)piperidine-3-carboxamide and 4-benzyl-N-(2,6-dimethylphenyl)piperazine-2-carboxamide, two conformationally restricted analogues of tocainide, were designed and synthesized as voltage-gated skeletal muscle sodium channel blockers... |
|
|
2D- and 3D-QSAR of tocainide and mexiletine analogues acting as Na(v)1.4 channel blockers.
Eur. J. Med. Chem. 44(4) , 1477-85, (2009) Enantiomeric forms of Tocainide, Mexiletine, and structurally related local anaesthetic compounds, were analyzed with respect to their potency in blocking Na(v)1.4 channel. Structure-activity relationships based on in vitro pharmacological assays, suggested t... |
|
|
New potent mexiletine and tocainide analogues evaluated in vivo and in vitro as antimyotonic agents on the myotonic ADR mouse.
Neuromuscul. Disord. 14(7) , 405-16, (2004) The antimyotonic activity of chiral derivatives of mexiletine and tocainide, selected as potent use-dependent blockers of skeletal muscle sodium channels, was evaluated in vivo acutely in myotonic ADR mice. The compounds had either aromatic (Me4 and Me6) or b... |