2D- and 3D-QSAR of tocainide and mexiletine analogues acting as Na(v)1.4 channel blockers.
Antonio Carrieri, Marilena Muraglia, Filomena Corbo, Concetta Pacifico, Antonio Carrieri, Marilena Muraglia, Filomena Corbo, Concetta Pacifico
Index: Eur. J. Med. Chem. 44(4) , 1477-85, (2009)
Full Text: HTML
Abstract
Enantiomeric forms of Tocainide, Mexiletine, and structurally related local anaesthetic compounds, were analyzed with respect to their potency in blocking Na(v)1.4 channel. Structure-activity relationships based on in vitro pharmacological assays, suggested that an increase in terms of lipophilicity and/or molecular surface as well as the presence of specific polar spacers might be determinant for receptor interactions. QSAR and pharmacophore models were then used to support at 3D level this hypothesis.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
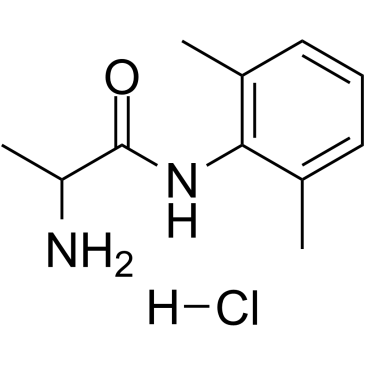 |
Tocainide hydrochloride
CAS:71395-14-7 |
C11H17ClN2O |
|
Synthesis, structure and pharmacology of acyl-2,6-xylidines.
2004-01-01 [Acta Pol. Pharm. 61(3) , 215-21, (2004)] |
|
Quantitative structure-pharmacokinetic relationships for dru...
2006-03-01 [J. Mol. Graph. Model. 24(5) , 383-95, (2006)] |
|
The efficacy of anticonvulsants on orofacial pain: a systema...
2011-05-01 [Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 111(5) , 627-33, (2011)] |
|
Non-antiepileptic drugs for trigeminal neuralgia.
2011-01-01 [Cochrane Database Syst. Rev. (1) , CD004029, (2011)] |
|
Tocainide analogues binding to human serum albumin: a HPLAC ...
2010-10-10 [J. Pharm. Biomed. Anal. 53(2) , 179-85, (2010)] |