(R)-Alcohol dehydrogenase
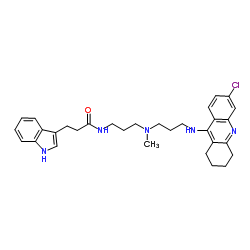
(R)-Alcohol dehydrogenase structure
|
Common Name | (R)-Alcohol dehydrogenase | ||
|---|---|---|---|---|
| CAS Number | 9028-12-0 | Molecular Weight | 532.119 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 806.8±65.0 °C at 760 mmHg | |
| Molecular Formula | C31H38ClN5O | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 441.7±34.3 °C | |
| Symbol |

GHS08 |
Signal Word | Danger | |
|
Efficient one-step production of (S)-1-phenyl-1,2-ethanediol from (R)-enantiomer plus NAD(+)-NADPH in-situ regeneration using engineered Escherichia coli.
Microb. Cell Fact. 11 , 167, (2012) Candida parapsilosis CCTCC M203011 catalyzes the stereoinversion of (R)-1-phenyl-1,2-ethanediol (PED) through oxidation and reduction. Its NAD(+)-linked (R)-carbonyl reductase (RCR) catalyzes the oxidization of (R)-PED to 2-hydroxyacetophenone (HAP), and its ... |
|
|
A new strategy to improve the efficiency and sustainability of Candida parapsilosis catalyzing deracemization of (R,S)-1-phenyl-1,2-ethanediol under non-growing conditions: increase of NADPH availability.
J. Microbiol. Biotechnol. 19(1) , 65-71, (2009) Microbial oxidoreductive systems have been widely used in asymmetric syntheses of optically active alcohols. However, when reused in multi-batch reaction, the catalytic efficiency and sustainability of non-growing cells usually decreased because of continuous... |
|
|
Stereospecificity of ketoreductase domains 1 and 2 of the tylactone modular polyketide synthase.
J. Am. Chem. Soc. 130 , 11598-11599, (2008) Tylactone synthase (TYLS) is a modular polyketide synthase that catalyzes the formation of tylactone (1), the parent aglycone precursor of the macrolide antibiotic tylosin. TYLS modules 1 and 2 are responsible for the generation of antidiketide and triketide ... |
|
|
Inhibition of carbonyl reductase activity in pig heart by alkyl phenyl ketones.
J. Enzyme Inhib. Med. Chem. 22(1) , 105-9, (2007) The inhibitory effects of alkyl phenyl ketones on carbonyl reductase activity were examined in pig heart. In this study, carbonyl reductase activity was estimated as the ability to reduce 4-benzoylpyridine to S(-)-alpha-phenyl-4-pyridylmethanol in the cytosol... |
|
|
De novo biosynthesis of vanillin in fission yeast (Schizosaccharomyces pombe) and baker's yeast (Saccharomyces cerevisiae).
Appl. Environ. Microbiol. 75(9) , 2765-74, (2009) Vanillin is one of the world's most important flavor compounds, with a global market of 180 million dollars. Natural vanillin is derived from the cured seed pods of the vanilla orchid (Vanilla planifolia), but most of the world's vanillin is synthesized from ... |
|
|
NADH- vs NADPH-coupled reduction of 5-hydroxymethyl furfural (HMF) and its implications on product distribution in Saccharomyces cerevisiae.
Appl. Microbiol. Biotechnol. 78(6) , 939-45, (2008) Saccharomyces cerevisiae alcohol dehydrogenases responsible for NADH-, and NADPH-specific reduction of the furaldehydes 5-hydroxymethyl-furfural (HMF) and furfural have previously been identified. In the present study, strains overexpressing the corresponding... |
|
|
Structure of aldehyde reductase holoenzyme in complex with the potent aldose reductase inhibitor fidarestat: implications for inhibitor binding and selectivity.
J. Med. Chem. 48(17) , 5536-42, (2005) Structure determination of porcine aldehyde reductase holoenzyme in complex with the potent aldose reductase inhibitor fidarestat was carried out to explain the difference in the potency of the inhibitor for aldose and aldehyde reductases. The hydrogen bonds ... |
|
|
Carbonyl reductase expression and its clinical significance in non-small-cell lung cancer.
Cancer Epidemiol. Biomarkers Prev. 14(8) , 1972-5, (2005) Carbonyl reductase (CBR) is a cytosolic NADPH-dependent oxidoreductase metabolizing prostaglandins, steroids, quinines, and anthracycline antibiotics. Many experimental studies have shown that CBR plays important roles in the regulation of tumor progression, ... |
|
|
Thermostable NADP(+)-dependent medium-chain alcohol dehydrogenase from Acinetobacter sp. strain M-1: purification and characterization and gene expression in Escherichia coli.
Appl. Environ. Microbiol. 66(12) , 5231-5, (2000) NADPH-dependent alkylaldehyde reducing enzyme, which was greatly induced by n-hexadecane, from Acinetobacter sp. strain M-1 was purified and characterized. The purified enzyme had molecular masses of 40 kDa as determined by sodium dodecyl sulfate-polyacrylami... |
|
|
Stereoselective synthesis of (R)-3-quinuclidinol through asymmetric reduction of 3-quinuclidinone with 3-quinuclidinone reductase of Rhodotorula rubra.
Appl. Microbiol. Biotechnol. 83(4) , 617-26, (2009) A novel nicotinamide adenine dinucleotide phosphate-dependent carbonyl reductase, 3-quinuclidinone reductase, was isolated from Rhodotorula rubra JCM3782. The enzyme catalyzes the asymmetric reduction of 3-quinuclidinone to (R)-3-quinuclidinol. The gene encod... |