| Structure | Name/CAS No. | Articles |
|---|---|---|
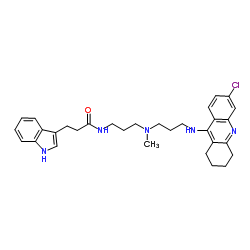 |
(R)-Alcohol dehydrogenase
CAS:9028-12-0 |
|
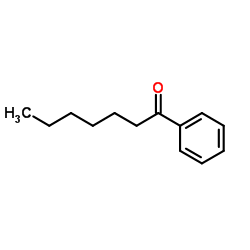 |
1-Phenyl-1-heptanone
CAS:1671-75-6 |