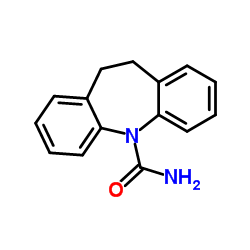
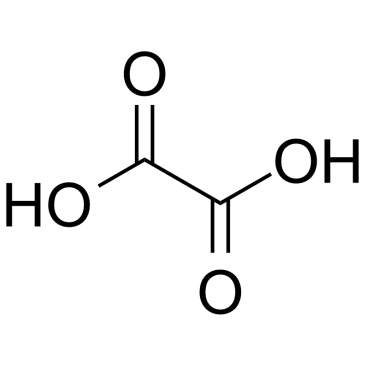
毒理学数据:
二、毒理学数据:
急性毒性:不可用。
生态学数据:
三、生态学数据:
1、其它有害作用:该物质对环境可能有危害,对水体应给予特别注意。
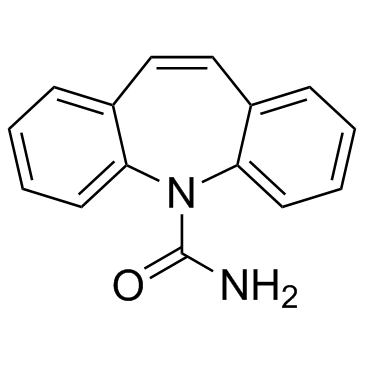
CHEMICAL IDENTIFICATION
-
RTECS NUMBER :
-
HN8225000
-
CHEMICAL NAME :
-
5H-Dibenz(b,f)azepine-5-carboxamide
-
CAS REGISTRY NUMBER :
-
298-46-4
-
BEILSTEIN REFERENCE NO. :
-
1246090
-
LAST UPDATED :
-
199706
-
DATA ITEMS CITED :
-
40
-
MOLECULAR FORMULA :
-
C15-H12-N2-O
-
MOLECULAR WEIGHT :
-
236.29
-
WISWESSER LINE NOTATION :
-
T C676 BNJ BVZ
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human - woman
-
DOSE/DURATION :
-
112 mg/kg/2W-I
-
TOXIC EFFECTS :
-
Gastrointestinal - nausea or vomiting Blood - thrombocytopenia
-
REFERENCE :
-
AJPSAO American Journal of Psychiatry. (American Psychiatric Assoc., Circulation Dept., 1400 K St., NW, Washington, DC 20005) V.78- 1921- Volume(issue)/page/year: 150,1750,1993
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human - child
-
DOSE/DURATION :
-
65 mg/kg
-
TOXIC EFFECTS :
-
Behavioral - convulsions or effect on seizure threshold Behavioral - coma Lungs, Thorax, or Respiration - other changes
-
REFERENCE :
-
JOPDAB Journal of Pediatrics. (C.V. Mosby Co., 11830 Westline Industrial Dr., St. Louis, MO 63141) V.1- 1932- Volume(issue)/page/year: 121,295,1992
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human - woman
-
DOSE/DURATION :
-
144 mg/kg/2W-I
-
TOXIC EFFECTS :
-
Blood - thrombocytopenia Skin and Appendages - dermatitis, allergic (after systemic exposure)
-
REFERENCE :
-
JCLPDE Journal of Clinical Psychiatry. (Physicians Postgraduate Press, Inc., POB 240008, Memphis, TN 38124) V.39- 1978- Volume(issue)/page/year: 53,378,1992
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human - man
-
DOSE/DURATION :
-
160 mg/kg/3W-I
-
TOXIC EFFECTS :
-
Skin and Appendages - dermatitis, other (after systemic exposure)
-
REFERENCE :
-
JCPYDR Journal of Clinical Pyschopharmacology. (Williams & Wilkins Co., 428 E. Preston St., Baltimore, MD 21202) V.1- 1981- Volume(issue)/page/year: 5,185,1985
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human - child
-
DOSE/DURATION :
-
1050 mg/kg/6W-I
-
TOXIC EFFECTS :
-
Behavioral - muscle contraction or spasticity
-
REFERENCE :
-
AJPSAO American Journal of Psychiatry. (American Psychiatric Assoc., Circulation Dept., 1400 K St., NW, Washington, DC 20005) V.78- 1921- Volume(issue)/page/year: 143,1176,1985
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human - man
-
DOSE/DURATION :
-
253 mg/kg/6W-I
-
TOXIC EFFECTS :
-
Gastrointestinal - nausea or vomiting Liver - liver function tests impaired
-
REFERENCE :
-
JCPYDR Journal of Clinical Pyschopharmacology. (Williams & Wilkins Co., 428 E. Preston St., Baltimore, MD 21202) V.1- 1981- Volume(issue)/page/year: 6,251,1986
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human - child
-
DOSE/DURATION :
-
19 mg/kg/4W-I
-
TOXIC EFFECTS :
-
Peripheral Nerve and Sensation - fasciculations
-
REFERENCE :
-
PEDIAU Pediatrics. (American Academy of Pediatrics, P.O. Box 1034, Evanston, IL 60204) V.1- 1948- Volume(issue)/page/year: 73,841,1984
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human - woman
-
DOSE/DURATION :
-
28 mg/kg/4D-I
-
TOXIC EFFECTS :
-
Behavioral - somnolence (general depressed activity) Gastrointestinal - nausea or vomiting Nutritional and Gross Metabolic - other changes
-
REFERENCE :
-
AJPSAO American Journal of Psychiatry. (American Psychiatric Assoc., Circulation Dept., 1400 K St., NW, Washington, DC 20005) V.78- 1921- Volume(issue)/page/year: 143,1328,1986
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human
-
DOSE/DURATION :
-
43 mg/kg
-
TOXIC EFFECTS :
-
Behavioral - sleep Behavioral - hallucinations, distorted perceptions Gastrointestinal - nausea or vomiting
-
REFERENCE :
-
BMJOAE British Medical Journal. (British Medical Assoc., BMA House, Tavistock Sq., London WC1H 9JR, UK) V.1- 1857- Volume(issue)/page/year: 1,754,1977
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human - woman
-
DOSE/DURATION :
-
560 mg/kg/4W-I
-
TOXIC EFFECTS :
-
Blood - changes in bone marrow (not otherwise specified)
-
REFERENCE :
-
BJCPAT British Journal of Clinical Practice. (Medical News Group, 1 Bedford St., London WC2E 9HD, UK) V.10(10)- 1956- Volume(issue)/page/year: 43,302,1989
-
TYPE OF TEST :
-
LDLo - Lowest published lethal dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human - man
-
DOSE/DURATION :
-
54 mg/kg/9D-I
-
TOXIC EFFECTS :
-
Behavioral - ataxia Kidney, Ureter, Bladder - urine volume increased Blood - agranulocytosis
-
REFERENCE :
-
CMAJAX Canadian Medical Association Journal. (Canadian Medical Assoc., POB 8650, Ottawa, ON K1G 0G8, Canada) V.1- 1911- Volume(issue)/page/year: 132,1040,1985
-
TYPE OF TEST :
-
LDLo - Lowest published lethal dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human - woman
-
DOSE/DURATION :
-
1920 mg/kg/17W-I
-
TOXIC EFFECTS :
-
Blood - aplastic anemia
-
REFERENCE :
-
AJPSAO American Journal of Psychiatry. (American Psychiatric Assoc., Circulation Dept., 1400 K St., NW, Washington, DC 20005) V.78- 1921- Volume(issue)/page/year: 142,974,1985
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human - man
-
DOSE/DURATION :
-
94 mg/kg/11D-I
-
TOXIC EFFECTS :
-
Blood - thrombocytopenia
-
REFERENCE :
-
JCPYDR Journal of Clinical Pyschopharmacology. (Williams & Wilkins Co., 428 E. Preston St., Baltimore, MD 21202) V.1- 1981- Volume(issue)/page/year: 10,305,1990
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
1957 mg/kg
-
TOXIC EFFECTS :
-
Behavioral - altered sleep time (including change in righting reflex)
-
REFERENCE :
-
JKXXAF Japanese Kokai Tokyo Koho Patents. (U.S. Patent and Trademark Office, Foreign Patents, Washington, DC 20231) Volume(issue)/page/year: #79-163823
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Intraperitoneal
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
158 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
REFERENCE :
-
AIPTAK Archives Internationales de Pharmacodynamie et de Therapie. (Heymans Institute of Pharmacology, De Pintelaan 185, B-9000 Ghent, Belgium) V.4- 1898- Volume(issue)/page/year: 202,106,1973
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Subcutaneous
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
>1500 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
REFERENCE :
-
ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag, Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951- Volume(issue)/page/year: 30,477,1980
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - mouse
-
DOSE/DURATION :
-
529 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
REFERENCE :
-
FRMCE8 Farmaco. (Societa Chimica Italiana, Corso Strada Nova, 86, Casella Postale 227, 27100 Pavia, Italy) V.44- 1989- Volume(issue)/page/year: 44,595,1989
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Intraperitoneal
-
SPECIES OBSERVED :
-
Rodent - mouse
-
DOSE/DURATION :
-
114 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
REFERENCE :
-
FATOAO Farmakologiya i Toksikologiya (Moscow). For English translation, see PHTXA6 and RPTOAN. (V/O Mezhdunarodnaya Kniga, 113095 Moscow, USSR) V.2- 1939- Volume(issue)/page/year: 53(4),19,1990
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Subcutaneous
-
SPECIES OBSERVED :
-
Rodent - mouse
-
DOSE/DURATION :
-
>1 gm/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
REFERENCE :
-
ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag, Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951- Volume(issue)/page/year: 30,477,1980
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Mammal - dog
-
DOSE/DURATION :
-
5620 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
REFERENCE :
-
27ZQAG "Psychotropic Drugs and Related Compounds," 2nd ed., Usdin, E., and D.H. Efron, Washington, DC, 1972 Volume(issue)/page/year: -,63,1972
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - rabbit
-
DOSE/DURATION :
-
2680 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
REFERENCE :
-
27ZQAG "Psychotropic Drugs and Related Compounds," 2nd ed., Usdin, E., and D.H. Efron, Washington, DC, 1972 Volume(issue)/page/year: -,63,1972
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - guinea pig
-
DOSE/DURATION :
-
920 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
REFERENCE :
-
27ZQAG "Psychotropic Drugs and Related Compounds," 2nd ed., Usdin, E., and D.H. Efron, Washington, DC, 1972 Volume(issue)/page/year: -,63,1972 ** REPRODUCTIVE DATA **
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
96 mg/kg
-
SEX/DURATION :
-
female 3 week(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Specific Developmental Abnormalities - Central Nervous System
-
REFERENCE :
-
OBGNAS Obstetrics and Gynecology. (Elsevier Science Pub. Co., Inc., 52 Vanderbilt Ave., New York, NY 10017) V.1- 1953- Volume(issue)/page/year: 82,705,1993
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
3192 mg/kg
-
SEX/DURATION :
-
female 1-38 week(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Specific Developmental Abnormalities - craniofacial (including nose and tongue) Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight gain) Reproductive - Effects on Newborn - delayed effects
-
REFERENCE :
-
NEJMAG New England Journal of Medicine. (Massachusetts Medical Soc., 10 Shattuck St., Boston, MA 02115) V.198- 1928- Volume(issue)/page/year: 320,1661,1989
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
7644 mg/kg
-
SEX/DURATION :
-
female 1-39 week(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Specific Developmental Abnormalities - skin and skin appendages Reproductive - Specific Developmental Abnormalities - cardiovascular (circulatory) system
-
REFERENCE :
-
NEJMAG New England Journal of Medicine. (Massachusetts Medical Soc., 10 Shattuck St., Boston, MA 02115) V.198- 1928- Volume(issue)/page/year: 320,1661,1989
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
1288 mg/kg
-
SEX/DURATION :
-
female 26-42 week(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Specific Developmental Abnormalities - skin and skin appendages
-
REFERENCE :
-
NRPDDB Neuropediatrics (Stuttgart). (Hippokrates Verlag GmbH, Postfach 593, D-7000 Stuttgart 1, Fed. Rep. Ger.) V.11- 1980- Volume(issue)/page/year: 16,167,1985
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
3492 mg/kg
-
SEX/DURATION :
-
female 1-39 week(s) after conception lactating female 17 day(s) post-birth
-
TOXIC EFFECTS :
-
Reproductive - Effects on Newborn - biochemical and metabolic
-
REFERENCE :
-
EJPEDT European Journal of Pediatrics. (Springer-Verlag New York, Inc., Service Center, 44 Hartz Way, Secaucus, NJ 07094) V.121- 1975- Volume(issue)/page/year: 150,136,1990
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
3600 mg/kg
-
SEX/DURATION :
-
female 14 day(s) pre-mating female 1-22 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Effects on Newborn - live birth index (measured after birth) Reproductive - Effects on Newborn - weaning or lactation index (e.g., # alive at weaning per # alive at day 4) Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight gain)
-
REFERENCE :
-
TJADAB Teratology, The International Journal of Abnormal Development. (Alan R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968- Volume(issue)/page/year: 29(3),33A,1984
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
765 mg/kg
-
SEX/DURATION :
-
female 9-17 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Fertility - litter size (e.g. # fetuses per litter; measured before birth) Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death, e.g., stunted fetus) Reproductive - Effects on Embryo or Fetus - fetal death
-
REFERENCE :
-
EAMJAV East African Medical Journal. (P.O. Box 41632, Nairobi, Kenya) V.9- 1932- Volume(issue)/page/year: 60,407,1983
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
4800 mg/kg
-
SEX/DURATION :
-
female 7-18 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Specific Developmental Abnormalities - other developmental abnormalities
-
REFERENCE :
-
TJADAB Teratology, The International Journal of Abnormal Development. (Alan R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968- Volume(issue)/page/year: 41,311,1990
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
7200 mg/kg
-
SEX/DURATION :
-
female 7-18 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Fertility - post-implantation mortality (e.g. dead and/or resorbed implants per total number of implants) Reproductive - Specific Developmental Abnormalities - musculoskeletal system
-
REFERENCE :
-
TJADAB Teratology, The International Journal of Abnormal Development. (Alan R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968- Volume(issue)/page/year: 41,311,1990
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
2400 mg/kg
-
SEX/DURATION :
-
female 7-18 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death, e.g., stunted fetus)
-
REFERENCE :
-
TJADAB Teratology, The International Journal of Abnormal Development. (Alan R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968- Volume(issue)/page/year: 41,311,1990
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
440 mg/kg
-
SEX/DURATION :
-
female 6-16 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
-
REFERENCE :
-
TXAPA9 Toxicology and Applied Pharmacology. (Academic Press, Inc., 1 E. First St., Duluth, MN 55802) V.1- 1959- Volume(issue)/page/year: 40,365,1977
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
92 gm/kg
-
SEX/DURATION :
-
male 2 week(s) pre-mating female 2 week(s) pre-mating - 18 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Fertility - pre-implantation mortality (e.g. reduction in number of implants per female; total number of implants per corpora lutea) Reproductive - Fertility - post-implantation mortality (e.g. dead and/or resorbed implants per total number of implants) Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death, e.g., stunted fetus)
-
REFERENCE :
-
TCMUD8 Teratogenesis, Carcinogenesis, and Mutagenesis. (Alan R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1980- Volume(issue)/page/year: 6,393,1986
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
5628 mg/kg
-
SEX/DURATION :
-
female 7-12 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Specific Developmental Abnormalities - eye/ear Reproductive - Specific Developmental Abnormalities - craniofacial (including nose and tongue)
-
REFERENCE :
-
TJADAB Teratology, The International Journal of Abnormal Development. (Alan R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968- Volume(issue)/page/year: 23,33A,1981
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
9984 mg/kg
-
SEX/DURATION :
-
female 8-13 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death, e.g., stunted fetus)
-
REFERENCE :
-
ARNEAS Archives of Neurology (Chicago). (AMA, 535 N. Dearborn St., Chicago, IL 60610) V.3- 1960- Volume(issue)/page/year: 36,832,1979 *** REVIEWS *** TOXICOLOGY REVIEW HFHBAG Hartford Hospital Bulletin. (Hartford Hospital, 80 Seymour St., Hartford, CT 06115) V.1- 1938/39- Volume(issue)/page/year: 30,31,1975 TOXICOLOGY REVIEW EPILAK Epilepsia (New York). (Raven Press, 1185 Ave. of the Americas, New York, NY 10036) Ser.1-3: 1909-55; Ser.4: V.1- 1959- Volume(issue)/page/year: 16,159,1975 *** NIOSH STANDARDS DEVELOPMENT AND SURVEILLANCE DATA *** NIOSH OCCUPATIONAL EXPOSURE SURVEY DATA : NOES - National Occupational Exposure Survey (1983) NOES Hazard Code - X4467 No. of Facilities: 169 (estimated) No. of Industries: 2 No. of Occupations: 5 No. of Employees: 5295 (estimated) No. of Female Employees: 3361 (estimated)
|










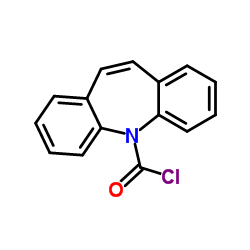
![5-氰基-5H-二苯并[b,f]氮杂卓结构式](https://image.chemsrc.com/caspic/033/42787-75-7.png)
![trans-10,11-dibromo-10,11-dihydro-5H-dibenz[b,f]azepine-5-carboxamide结构式](https://image.chemsrc.com/caspic/385/59690-99-2.png)