RO-3306
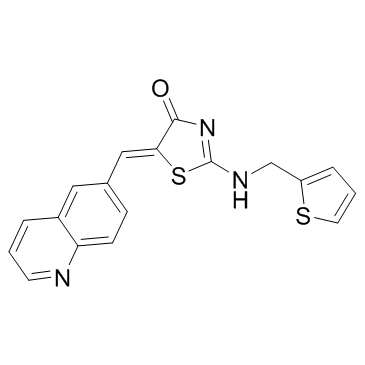
RO-3306 structure
|
Common Name | RO-3306 | ||
|---|---|---|---|---|
| CAS Number | 872573-93-8 | Molecular Weight | 351.445 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 569.1±60.0 °C at 760 mmHg | |
| Molecular Formula | C18H13N3OS2 | Melting Point | N/A | |
| MSDS | USA | Flash Point | 298.0±32.9 °C | |
Use of RO-3306Ro-3306 is a potent and selective inhibitor of CDK1, with Kis of 20 nM, 35 nM and 340 nM for CDK1, CDK1/cyclin B1 and CDK2/cyclin E, respectively. |
| Name | Ro-3306 |
|---|---|
| Synonym | More Synonyms |
| Description | Ro-3306 is a potent and selective inhibitor of CDK1, with Kis of 20 nM, 35 nM and 340 nM for CDK1, CDK1/cyclin B1 and CDK2/cyclin E, respectively. |
|---|---|
| Related Catalog | |
| Target |
CDK1:20 nM (Ki) CDK1/cyclinB1:35 nM (Ki) CDK1/cyclin A:110 nM (Ki) CDK2/cyclinE:340 nM (Ki) PKCδ:318 nM (Ki) SGK:497 nM (Ki) ERK:1980 nM (Ki) |
| In Vitro | RO-3306 is an ATP-competitive inhibitor, and inhibits CDK1/cyclin A complexes with Ki of 110 nM. RO-3306 blocks the cell cycle in the G2/M phase of human cancer cells. RO-3306 (4 μM) induces apoptosis in cancer cells[1]. RO-3306 (5 μM) induces G2/M-phase cell cycle arrest and apoptosis of AML cells in a time-dependent manner. RO-3306 treatment significantly increases the percentage of Annexin V-positive cells in G1-phase cells without affecting the cell cycle distribution. RO-3306 enhances p53-mediated apoptosis. RO-3306 cooperates with Nutlin-3 in activating Bax and inducing mitochondrial apoptosis. RO-3306 (5 μM) downregulates antiapoptotic p21, Bcl-2 and survivin protein expression in AML. RO-3306 inhibits p53-induced p21 synthesis. RO-3306 does not inhibit RNA polymerase II CTD phosphorylation[2]. RO-3306 (10 μM) effectively arrests oocyte maturation. RO-3306 reduces the blastocyst formation in oocytes[3]. |
| Kinase Assay | The CDK assays are run by using recombinant human CDK/cyclin complexes (CDK1/cyclin B1, CDK1/cyclin A, CDK2/cyclin E, and CDK4/cyclin D) expressed and isolated from Hi5 insect cells. GST-cyclin B1, CDK1, GST-cyclin-E, CDK2, GST-CDK4, and cyclin D, are used in the assay. The GST-tagged proteins are coexpressed and purified in complex with their partners. All assays use a His-6-tagged fragment of pRB (amino acids 385-928) as a substrate. The protein is expressed from a construct. It is expressed in M15 Escherichia coli cells and bound on a Ni-chalated agarose column pretreated with 1 mM imidazole and eluted with 500 mM imidazole. The eluted protein is dialyzed against 20 mM Hepes, pH 7/6.25 mM MgCl2/1.5 mM DTT, aliquoted, and stored at −80°C. |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 569.1±60.0 °C at 760 mmHg |
| Molecular Formula | C18H13N3OS2 |
| Molecular Weight | 351.445 |
| Flash Point | 298.0±32.9 °C |
| Exact Mass | 351.049988 |
| PSA | 111.38000 |
| LogP | 3.24 |
| Appearance of Characters | white to light brown |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C |
| Index of Refraction | 1.746 |
| Storage condition | 2-8°C |
| Water Solubility | DMSO: soluble5mg/mL, clear |
| RIDADR | NONH for all modes of transport |
|---|
|
CDK1 substitutes for mTOR kinase to activate mitotic cap-dependent protein translation.
Proc. Natl. Acad. Sci. U. S. A. 112 , 5875-82, (2015) Mitosis is commonly thought to be associated with reduced cap-dependent protein translation. Here we show an alternative control mechanism for maintaining cap-dependent translation during mitosis reve... |
|
|
JNK2 controls fragmentation of the Golgi complex and the G2/M transition through phosphorylation of GRASP65.
J. Cell Sci. 128 , 2249-60, (2015) In mammalian cells, the Golgi complex is composed of stacks that are connected by membranous tubules. During G2, the Golgi complex is disassembled into isolated stacks. This process is required for en... |
|
|
A Cholesterol-Dependent Endocytic Mechanism Generates Midbody Tubules During Cytokinesis.
Traffic 16 , 1174-92, (2015) Cytokinesis is the final stage of cell division and produces two independent daughter cells. Vesicles derived from internal membrane stores, such as the Golgi, lysosomes, and early and recycling endos... |
| 4(5H)-Thiazolone, 5-(6-quinolinylmethylene)-2-[(2-thienylmethyl)amino]-, (5Z)- |
| (5Z)-5-(quinolin-6-ylmethylidene)-2-[(thiophen-2-ylmethyl)amino]-1,3-thiazol-4(5H)-one |
| (5Z)-5-(quinolin-6-ylmethylidene)-2-(thiophen-2-ylmethylamino)-1,3-thiazol-4-one |
| (5Z)-5-(6-Quinolinylmethylene)-2-[(2-thienylmethyl)amino]-1,3-thiazol-4(5H)-one |