CHEMICAL IDENTIFICATION
-
RTECS NUMBER :
-
UP0791400
-
CHEMICAL NAME :
-
6H-Purin-6-one, 1,9-dihydro-2-amino-9-((2-hydroxyethoxy)methyl)-
-
CAS REGISTRY NUMBER :
-
59277-89-3
-
LAST UPDATED :
-
199712
-
DATA ITEMS CITED :
-
30
-
MOLECULAR FORMULA :
-
C8-H11-N5-O3
-
MOLECULAR WEIGHT :
-
225.24
-
WISWESSER LINE NOTATION :
-
T56 BN DN FN HNJ D1O2Q GZ IQ &T56 BN DN FVM INJ B1O2Q HZ
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human - woman
-
DOSE/DURATION :
-
28 mg/kg/2D-I
-
TOXIC EFFECTS :
-
Brain and Coverings - changes in surface EEG Behavioral - hallucinations, distorted perceptions Lungs, Thorax, or Respiration - sputum
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human - woman
-
DOSE/DURATION :
-
12 mg/kg/1D-I
-
TOXIC EFFECTS :
-
Brain and Coverings - meningeal changes Behavioral - somnolence (general depressed activity) Behavioral - antipsychotic
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human - woman
-
DOSE/DURATION :
-
100 mg/kg/5D-I
-
TOXIC EFFECTS :
-
Skin and Appendages - dermatitis, allergic (after systemic exposure)
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human - woman
-
DOSE/DURATION :
-
80 mg/kg/4D-I
-
TOXIC EFFECTS :
-
Behavioral - somnolence (general depressed activity) Behavioral - hallucinations, distorted perceptions Behavioral - convulsions or effect on seizure threshold
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Human - man
-
DOSE/DURATION :
-
486 mg/kg/17D-I
-
TOXIC EFFECTS :
-
Gastrointestinal - decreased motility or constipation Gastrointestinal - other changes
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Intravenous
-
SPECIES OBSERVED :
-
Human - man
-
DOSE/DURATION :
-
134 ug/kg/1D-I
-
TOXIC EFFECTS :
-
Behavioral - hallucinations, distorted perceptions
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Intravenous
-
SPECIES OBSERVED :
-
Human - woman
-
DOSE/DURATION :
-
101 mg/kg/2D-I
-
TOXIC EFFECTS :
-
Behavioral - hallucinations, distorted perceptions Behavioral - muscle contraction or spasticity
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Intravenous
-
SPECIES OBSERVED :
-
Human - man
-
DOSE/DURATION :
-
107 mg/kg/4D-I
-
TOXIC EFFECTS :
-
Behavioral - hallucinations, distorted perceptions Kidney, Ureter, Bladder - changes in tubules (including acute renal failure, acute tubular necrosis)
-
TYPE OF TEST :
-
LDLo - Lowest published lethal dose
-
ROUTE OF EXPOSURE :
-
Multiple routes
-
SPECIES OBSERVED :
-
Human - woman
-
DOSE/DURATION :
-
36 mg/kg/2D-I
-
TOXIC EFFECTS :
-
Brain and Coverings - other degenerative changes Behavioral - coma Lungs, Thorax, or Respiration - other changes
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
>20 gm/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Intraperitoneal
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
860 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Subcutaneous
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
620 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Intravenous
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
750 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - mouse
-
DOSE/DURATION :
-
>10 gm/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Intraperitoneal
-
SPECIES OBSERVED :
-
Rodent - mouse
-
DOSE/DURATION :
-
724 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Subcutaneous
-
SPECIES OBSERVED :
-
Rodent - mouse
-
DOSE/DURATION :
-
1118 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Intravenous
-
SPECIES OBSERVED :
-
Rodent - mouse
-
DOSE/DURATION :
-
1118 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Intravenous
-
SPECIES OBSERVED :
-
Mammal - species unspecified
-
DOSE/DURATION :
-
400 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Intraperitoneal
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
250 mg/kg/5D-I
-
TOXIC EFFECTS :
-
Kidney, Ureter, Bladder - changes primarily in glomeruli Kidney, Ureter, Bladder - urine volume increased Nutritional and Gross Metabolic - weight loss or decreased weight gain
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
60 mg/kg
-
SEX/DURATION :
-
female 5-6 week(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Specific Developmental Abnormalities - Central Nervous System
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Subcutaneous
-
DOSE :
-
100 mg/kg
-
SEX/DURATION :
-
female 10 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Specific Developmental Abnormalities - musculoskeletal system Reproductive - Effects on Newborn - weaning or lactation index (e.g., # alive at weaning per # alive at day 4)
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Subcutaneous
-
DOSE :
-
200 mg/kg
-
SEX/DURATION :
-
female 10 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Specific Developmental Abnormalities - craniofacial (including nose and tongue)
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Subcutaneous
-
DOSE :
-
400 mg/kg
-
SEX/DURATION :
-
female 9-11 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death, e.g., stunted fetus)
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Subcutaneous
-
DOSE :
-
1200 mg/kg
-
SEX/DURATION :
-
female 1-20 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Maternal Effects - other effects Reproductive - Fertility - post-implantation mortality (e.g. dead and/or resorbed implants per total number of implants) Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death, e.g., stunted fetus)
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Unreported
-
DOSE :
-
300 mg/kg
-
SEX/DURATION :
-
female 10 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death, e.g., stunted fetus)
MUTATION DATA
-
TYPE OF TEST :
-
DNA inhibition
-
TEST SYSTEM :
-
Rodent - rabbit Kidney
-
DOSE/DURATION :
-
6800 ug/L
-
REFERENCE :
-
BCPCA6 Biochemical Pharmacology. (Pergamon Press Inc., Maxwell House, Fairview Park, Elmsford, NY 10523) V.1- 1958- Volume(issue)/page/year: 38,1771,1989
|
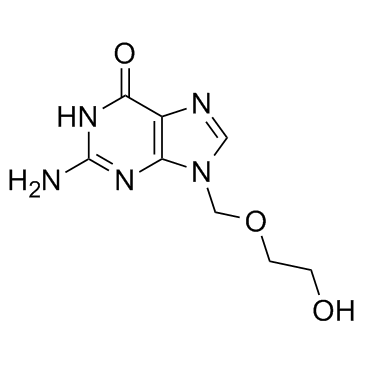
![9-[(2-Acetoxyethoxy)methyl]-acetylguanine Structure](https://www.chemsrc.com/caspic/238/75128-73-3.png) CAS#:75128-73-3
CAS#:75128-73-3 CAS#:646-06-0
CAS#:646-06-0 CAS#:73-40-5
CAS#:73-40-5 CAS#:7440-44-0
CAS#:7440-44-0![9-[(2-acetoxyethoxy)methyl]-2-(acetylamino)-6-(diphenylcarbamoyloxy)-9H-purine Structure](https://www.chemsrc.com/caspic/253/112233-78-0.png) CAS#:112233-78-0
CAS#:112233-78-0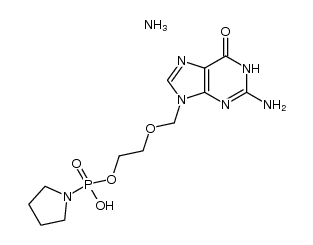 CAS#:1400637-35-5
CAS#:1400637-35-5![L-VALINE,N-[(1,2-DIMETHYLETHOXY)CARBONYL]-2-[(2-AMINO-1,6-DIHYDRO-6-OXO-9H-PURIN-9-YL)METHYOXY]ETHYLESTER structure](https://www.chemsrc.com/caspic/093/502421-44-5.png) CAS#:502421-44-5
CAS#:502421-44-5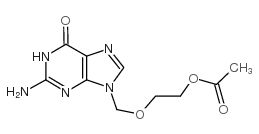 CAS#:102728-64-3
CAS#:102728-64-3![N2-Acetyl-9-[(2-hydroxyethoxy)methyl]guanine structure](https://www.chemsrc.com/caspic/332/110104-37-5.png) CAS#:110104-37-5
CAS#:110104-37-5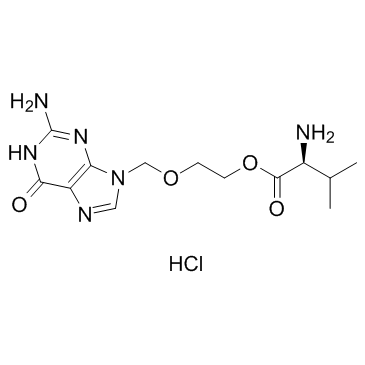 CAS#:124832-27-5
CAS#:124832-27-5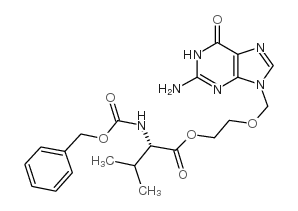 CAS#:124832-31-1
CAS#:124832-31-1![3-(2-hydroxyethoxymethyl)-6-methyl-5H-imidazo[1,2-a]purin-9-one structure](https://www.chemsrc.com/caspic/207/114199-19-8.png) CAS#:114199-19-8
CAS#:114199-19-8![3-(2-hydroxyethoxymethyl)-5,6-dimethylimidazo[1,2-a]purin-9-one structure](https://www.chemsrc.com/caspic/342/114199-20-1.png) CAS#:114199-20-1
CAS#:114199-20-1![3-(2-hydroxyethoxymethyl)-1,6-dimethylimidazo[1,2-a]purin-3-ium-9-olate structure](https://www.chemsrc.com/caspic/304/114199-21-2.png) CAS#:114199-21-2
CAS#:114199-21-2
