5a-androstane
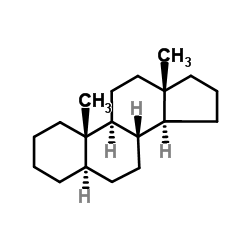
5a-androstane structure
|
Common Name | 5a-androstane | ||
|---|---|---|---|---|
| CAS Number | 438-22-2 | Molecular Weight | 260.457 | |
| Density | 0.9±0.1 g/cm3 | Boiling Point | 336.0±9.0 °C at 760 mmHg | |
| Molecular Formula | C19H32 | Melting Point | 78-82 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 145.9±12.3 °C | |
| Symbol |


GHS07, GHS08 |
Signal Word | Warning | |
| Name | 5α-androstane |
|---|---|
| Synonym | More Synonyms |
| Density | 0.9±0.1 g/cm3 |
|---|---|
| Boiling Point | 336.0±9.0 °C at 760 mmHg |
| Melting Point | 78-82 °C(lit.) |
| Molecular Formula | C19H32 |
| Molecular Weight | 260.457 |
| Flash Point | 145.9±12.3 °C |
| Exact Mass | 260.250397 |
| LogP | 8.42 |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.508 |
| Storage condition | -20℃ |
| Symbol |


GHS07, GHS08 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335-H336-H351-H373 |
| Precautionary Statements | P261-P281-P305 + P351 + P338 |
| Target Organs | Blood, Central nervous system, Liver |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
| Hazard Codes | T: Toxic; |
| Risk Phrases | 45-23/24/25-36/37/38-43-63 |
| Safety Phrases | S53-S23-S24/25-S36/37 |
| RIDADR | UN 1593 6.1/PG 3 |
| WGK Germany | 3 |
|
~% 
5a-androstane CAS#:438-22-2 |
| Literature: Butenandt; Tscherning Hoppe-Seyler's Zeitschrift fuer Physiologische Chemie, 1934 , vol. 229, p. 185,190 Full Text Show Details Heard; McKay Journal of Biological Chemistry, 1939 , vol. 131, p. 371,378 Full Text Show Details Reichstein Helvetica Chimica Acta, 1936 , vol. 19, p. 979,983 |
|
~% 
5a-androstane CAS#:438-22-2 |
| Literature: Burrows et al. Biochemical Journal, 1937 , vol. 31, p. 950,957 |
|
~% 
5a-androstane CAS#:438-22-2 |
| Literature: Burrows et al. Biochemical Journal, 1937 , vol. 31, p. 950,957 |
|
~% 
5a-androstane CAS#:438-22-2 |
| Literature: Burrows et al. Biochemical Journal, 1937 , vol. 31, p. 950,957 |
|
~% 
5a-androstane CAS#:438-22-2 |
| Literature: Butenandt; Tscherning Hoppe-Seyler's Zeitschrift fuer Physiologische Chemie, 1934 , vol. 229, p. 185,190 |
|
~% 
5a-androstane CAS#:438-22-2 |
| Literature: Kaegi; Miescher Helvetica Chimica Acta, 1939 , vol. 22, p. 683,695 Full Text Show Details Prelog; Ruzicka; Wieland Helvetica Chimica Acta, 1944 , vol. 27, p. 66,69 |
|
~% 
5a-androstane CAS#:438-22-2 |
| Literature: Reichstein Helvetica Chimica Acta, 1936 , vol. 19, p. 979,983 |
| Precursor 9 | |
|---|---|
| DownStream 0 | |
|
Potent and selective steroidal inhibitors of 17beta-hydroxysteroid dehydrogenase type 7, an enzyme that catalyzes the reduction of the key hormones estrone and dihydrotestosterone.
J. Med. Chem. 52 , 7488-502, (2009) 17beta-Hydroxysteroid dehydrogenase type 7 (17beta-HSD7) catalyzes the reduction of estrone (E(1)) into estradiol (E(2)) and of dihydrotestosterone (DHT) into 5alpha-androstane-3beta,17beta-diol (3bet... |
|
|
Novel and efficient synthesis and antifungal evaluation of 2,3-functionalized cholestane and androstane derivatives
Bioorg. Med. Chem. Lett. 20 , 7372-5, (2010) Synthetic modifications of cholesterol and other traditional steroid molecules have become a promising area for the exploration and development of novel antifungal agents, especially with respect to t... |
|
|
[Bioconversion of C19- and C21-steroids with parent and mutant strains of Curvularia lunata].
Prikl. Biokhim. Mikrobiol. 46(2) , 212-20, (2010) Regio- and stereospecificity of microbial hydroxylation was studied at the transformation of 3-keto-4-ene steroids of androstane and pregnane series by the filamentous fungus of Curvularia lunata VKMF... |
| Aetioallocholane |
| 5α-Androstan |
| 5alpha-Androstane |
| MFCD00067600 |
| (5a)-Androstane |
| (5α)-Androstane |
| Etioallocholane |
| (5R,8S,9S,10S,13S,14S)-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthrene |
| 5α,14α-Androstane |
| 5a-androstane |
| EINECS 207-116-2 |
| Aetioallocholan |
| Androstane, (5α)- |
| 5α-androstane |
| Androstane |