GR 89696 fumarate
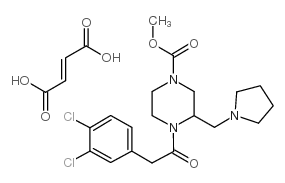
GR 89696 fumarate structure
|
Common Name | GR 89696 fumarate | ||
|---|---|---|---|---|
| CAS Number | 126766-32-3 | Molecular Weight | 530.39800 | |
| Density | N/A | Boiling Point | 559.2ºC at 760 mmHg | |
| Molecular Formula | C23H29Cl2N3O7 | Melting Point | N/A | |
| MSDS | USA | Flash Point | 292ºC | |
Use of GR 89696 fumarateGR 89696 is a highly selective κ2 opioid receptor agonist with potential to prevent pruritus[1]. |
| Name | GR 89696 fumarate,4-[(3,4-Dichlorophenyl)acetyl]-3-(1-pyrrolidinylmethyl)-1-piperazinecarboxylicacidmethylesterfumarate |
|---|
| Description | GR 89696 is a highly selective κ2 opioid receptor agonist with potential to prevent pruritus[1]. |
|---|---|
| Related Catalog | |
| In Vivo | GR 89696 (intramuscular injection, 0.01-0.1 μg/kg) 可以以一种剂量依赖的方式减轻鞘内注射吗啡(0.03mg)诱导的抓挠反应而不影响吗啡镇痛作用[1]。 GR-896960(subcutaneous injection, 1 mg/kg) 在大鼠永久性局灶性缺血模型中,使大脑动脉梗塞体积减少了 38%[2]。 |
| References |
| Boiling Point | 559.2ºC at 760 mmHg |
|---|---|
| Molecular Formula | C23H29Cl2N3O7 |
| Molecular Weight | 530.39800 |
| Flash Point | 292ºC |
| Exact Mass | 529.13800 |
| PSA | 127.69000 |
| LogP | 2.43640 |
| Vapour Pressure | 1.55E-12mmHg at 25°C |
| InChIKey | ABTNETSDXZBJTE-WLHGVMLRSA-N |
| SMILES | COC(=O)N1CCN(C(=O)Cc2ccc(Cl)c(Cl)c2)C(CN2CCCC2)C1.O=C(O)C=CC(=O)O |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |
|
[11C]GR103545: novel one-pot radiosynthesis with high specific activity.
Nucl. Med. Biol. 38(2) , 215-21, (2011) GR103545 is a potent and selective kappa-opioid receptor agonist. Previous studies in non-human primates demonstrated favorable properties of [(11)C]GR103545 as a positron emission tomography tracer f... |
|
|
GR89,696: a potent kappa-opioid agonist with subtype selectivity in rhesus monkeys.
J. Pharmacol. Exp. Ther. 298(3) , 1049-59, (2001) GR89,696 is a synthetic kappa-opioid receptor agonist, recently reported to have an agonist profile consistent with selectivity at the proposed "kappa(2)" subtype. The present studies evaluated the ef... |
|
|
[(11)C]-GR89696, a potent kappa opiate receptor radioligand; in vivo binding of the R and S enantiomers.
Nucl. Med. Biol. 29(1) , 47-53, (2002) The R and S enantiomers of [(11)C]GR89696, [(11)C]-methyl 4-[(3,4-dichlorophenyl)acetyl]-3-[(1-pyrrolidinyl)methyl]-1-piperazinecarboxylate, were synthesized from their appropriate chiral precursors a... |