Penicillin G potassium
Modify Date: 2024-01-02 19:50:07
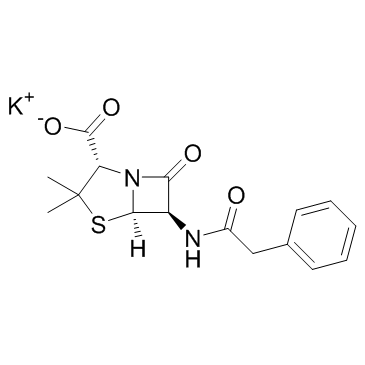
Penicillin G potassium structure
|
Common Name | Penicillin G potassium | ||
|---|---|---|---|---|
| CAS Number | 113-98-4 | Molecular Weight | 372.480 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C16H17KN2O4S | Melting Point | 214-217 ℃ | |
| MSDS | Chinese USA | Flash Point | N/A | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of Penicillin G potassiumPenicillin G potassium is a fast-acting antibiotic; used to treat bacterial infections that affect the blood, heart, lungs, joints, and genital areas. |
| Name | benzylpenicillin potassium |
|---|---|
| Synonym | More Synonyms |
| Description | Penicillin G potassium is a fast-acting antibiotic; used to treat bacterial infections that affect the blood, heart, lungs, joints, and genital areas. |
|---|---|
| Related Catalog | |
| References |
| Melting Point | 214-217 ℃ |
|---|---|
| Molecular Formula | C16H17KN2O4S |
| Molecular Weight | 372.480 |
| Exact Mass | 372.054596 |
| PSA | 114.84000 |
| Index of Refraction | 294 ° (C=1, H2O) |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H317 |
| Precautionary Statements | P280 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Faceshields;Gloves |
| Hazard Codes | Xn |
| Risk Phrases | 42/43-34-11 |
| Safety Phrases | 36/37-45-36/37/39-26-16 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 2 |
| RTECS | XH9700000 |
| HS Code | 2941109900 |
|
~96% 
Penicillin G po... CAS#:113-98-4 |
| Literature: WO2007/63107 A1, ; Page/Page column 7-8 ; |
|
~94% 
Penicillin G po... CAS#:113-98-4 |
| Literature: Journal of Organic Chemistry, , vol. 47, # 3 p. 587 - 590 |
|
~93% 
Penicillin G po... CAS#:113-98-4 |
| Literature: Journal of Organic Chemistry, , vol. 47, # 3 p. 587 - 590 |
| HS Code | 2941109900 |
|---|
|
The inducers 1,3-diaminopropane and spermidine produce a drastic increase in the expression of the penicillin biosynthetic genes for prolonged time, mediated by the laeA regulator.
Fungal Genet. Biol. 49(12) , 1004-13, (2012) We described previously that an autoinducer molecule, identified as 1,3-diaminopropane (1,3-DAP), is secreted by Penicillium chrysogenum and Acremonium chrysogenum. Using pH-controlled fermentor cultu... |
|
|
Chronic inflammatory demyelinating polyneuropathy associated with neurosyphilis.
Am. J. Med. Sci. 349(1) , 90-1, (2015)
|
|
|
[Clinical toxicology of mushroom poisoning. Amanita virosa].
Chudoku. Kenkyu. 26(3) , 210-4, (2013)
|
| Benzyl Penicillinate Potassium Salt |
| MFCD00036193 |
| Benzylpenicillinic acid potassium salt |
| Forpen |
| Potassium (1E)-N-[(2S,5R,6R)-2-carboxy-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]hept-6-yl]-2-phenylethanimidate |
| Benzylpenicillin potassium |
| Potassium (2S,5R,6R)-3,3-dimethyl-7-oxo-6-[(phenylacetyl)amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate |
| Tabilin |
| Hipercilina |
| Hyasorb |
| Penicillin G potassium |
| Benzylpenicillin Potassium Salt |
| Monopen |
| Eskacillin |
| 4-Thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid, 6-[[(1E)-1-hydroxy-2-phenylethylidene]amino]-3,3-dimethyl-7-oxo-, potassium salt, (2S,5R,6R)- (1:1) |
| [2S-(2a,5a,6b)]-3,3-Dimethyl-7-oxo-6-[(phenylacetyl)amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic Acid Monopotassium Salt |
| POTASSIUM PENICILLIN G |
| Hylenta |
| Notaral |
| 4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid, 3,3-dimethyl-7-oxo-6-[(phenylacetyl)amino]-, monopotassium salt, (2S,5R,6R)- |
| Pentid |
| Potassium benzylpenicillinate |
| potassium,(2S,5R,6R)-3,3-dimethyl-7-oxo-6-[(2-phenylacetyl)amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate |
| EINECS 204-038-0 |
| Scotcil |
| Penicillin G potassium salt |
| 4-Thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid, 3,3-dimethyl-7-oxo-6-[(2-phenylacetyl)amino]-, potassium salt, (2S,5R,6R)- (1:1) |
| Penicillin G (potassium) |

 CAS#:52-66-4
CAS#:52-66-4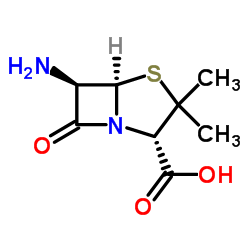 CAS#:551-16-6
CAS#:551-16-6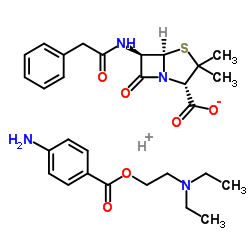 CAS#:54-35-3
CAS#:54-35-3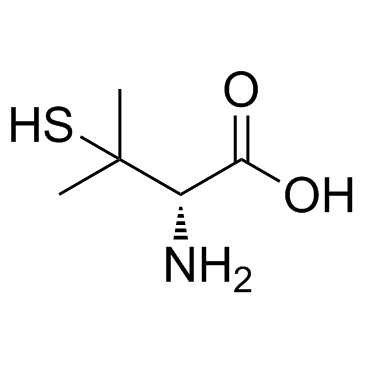 CAS#:52-67-5
CAS#:52-67-5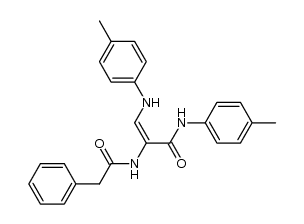 CAS#:124827-15-2
CAS#:124827-15-2 CAS#:51-17-2
CAS#:51-17-2 CAS#:621-72-7
CAS#:621-72-7
