Sumatriptan succinate
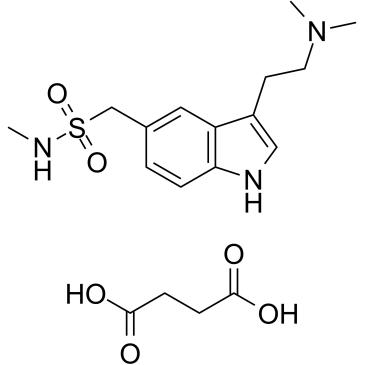
Sumatriptan succinate structure
|
Common Name | Sumatriptan succinate | ||
|---|---|---|---|---|
| CAS Number | 103628-48-4 | Molecular Weight | 341.426 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 497.7ºC at 760 mmHg | |
| Molecular Formula | C18H27N3O6S | Melting Point | 165-166°C | |
| MSDS | USA | Flash Point | 254.8ºC | |
| Symbol |

GHS05 |
Signal Word | Danger | |
Use of Sumatriptan succinateSumatriptan succinate (GR 43175) is a serotonin1 (5-HT1) receptor agonist, which is effective in the acute treatment of migraine headache.Target: 5-HT 1d receptor agonistSumatriptan succinate is a serotonin1 (5-HT1) receptor agonist, which is effective in the acute treatment of migraine headache. Its antimigraine activity is believed to derive from selective vasoconstriction of cranial blood vessels which are dilated and distended during migraine headache and/or from inhibition of neurogenically mediated inflammation in the dura mater [1].For sumatriptan succinate 50 mg versus placebo the NNTs were 6.1, 7.5, and 4.0 for pain-free at two hours and headache relief at one and two hours, respectively. NNTs for sustained pain-free and sustained headache relief during the 24 hours postdose were 9.5 and 6.0, respectively [2]. Difference in time-weighted (0-2.5 h) mean arterial pressure MAP (90% confidence interval) was 1.2 mmHg (-0.2, 2.7) between telcagepant and placebo, 4.0 mmHg (2.5, 5.5) between sumatriptan succinate and placebo, and 1.5 mmHg (0.0, 3.0) between telcagepant with sumatriptan succinate vs sumatriptan succinate alone. When coadministered with telcagepant, the AUC0-6h and C(max) of sumatriptan succinate were increased by 23% and 24%, respectively. The small MAP increases observed after coadministration could possibly be associated with the slight elevations in sumatriptan succinate levels [3].Clinical indications: Cluster headache; MigraineToxicity: Symptoms of overdose include convulsions, tremor, paralysis, inactivity, ptosis, erythema of the extremities, abnormal respiration, cyanosis, ataxia, mydriasis, salivation, and lacrimation |
| Name | sumatriptan succinate |
|---|---|
| Synonym | More Synonyms |
| Description | Sumatriptan succinate (GR 43175) is a serotonin1 (5-HT1) receptor agonist, which is effective in the acute treatment of migraine headache.Target: 5-HT 1d receptor agonistSumatriptan succinate is a serotonin1 (5-HT1) receptor agonist, which is effective in the acute treatment of migraine headache. Its antimigraine activity is believed to derive from selective vasoconstriction of cranial blood vessels which are dilated and distended during migraine headache and/or from inhibition of neurogenically mediated inflammation in the dura mater [1].For sumatriptan succinate 50 mg versus placebo the NNTs were 6.1, 7.5, and 4.0 for pain-free at two hours and headache relief at one and two hours, respectively. NNTs for sustained pain-free and sustained headache relief during the 24 hours postdose were 9.5 and 6.0, respectively [2]. Difference in time-weighted (0-2.5 h) mean arterial pressure MAP (90% confidence interval) was 1.2 mmHg (-0.2, 2.7) between telcagepant and placebo, 4.0 mmHg (2.5, 5.5) between sumatriptan succinate and placebo, and 1.5 mmHg (0.0, 3.0) between telcagepant with sumatriptan succinate vs sumatriptan succinate alone. When coadministered with telcagepant, the AUC0-6h and C(max) of sumatriptan succinate were increased by 23% and 24%, respectively. The small MAP increases observed after coadministration could possibly be associated with the slight elevations in sumatriptan succinate levels [3].Clinical indications: Cluster headache; MigraineToxicity: Symptoms of overdose include convulsions, tremor, paralysis, inactivity, ptosis, erythema of the extremities, abnormal respiration, cyanosis, ataxia, mydriasis, salivation, and lacrimation |
|---|---|
| Related Catalog | |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 497.7ºC at 760 mmHg |
| Melting Point | 165-166°C |
| Molecular Formula | C18H27N3O6S |
| Molecular Weight | 341.426 |
| Flash Point | 254.8ºC |
| Exact Mass | 341.140930 |
| PSA | 148.18000 |
| LogP | 0.34 |
| Vapour Pressure | 1.24E-17mmHg at 25°C |
| Index of Refraction | 1.552 |
| InChIKey | PORMUFZNYQJOEI-UHFFFAOYSA-N |
| SMILES | CNS(=O)(=O)Cc1ccc2[nH]cc(CCN(C)C)c2c1.O=C(O)CCC(=O)O |
| Storage condition | Store at +4°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS05 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H318 |
| Precautionary Statements | P280-P305 + P351 + P338 + P310 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi: Irritant; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S16-S26-S36-S61-S60 |
| RIDADR | UN 1648 3/PG 2 |
| HS Code | 2935009090 |
|
~% 
Sumatriptan suc... CAS#:103628-48-4 |
| Literature: WO2003/105836 A1, ; Page 17 ; |
|
~% 
Sumatriptan suc... CAS#:103628-48-4 |
| Literature: US2004/143002 A1, ; Page 2-3 ; |
| Precursor 2 | |
|---|---|
| DownStream 1 | |
| HS Code | 2935009090 |
|---|---|
| Summary | 2935009090 other sulphonamides VAT:17.0% Tax rebate rate:9.0% Supervision conditions:none MFN tariff:6.5% General tariff:35.0% |
|
Multidrug toxicity involving sumatriptan.
J. Anal. Toxicol. 39(1) , 75-9, (2015) A multidrug fatality involving sumatriptan is reported. Sumatriptan is a tryptamine derivative that acts at 5-HT(1B/1D) receptors and is used for the treatment of migraines. The decedent was a 21-year... |
|
|
Thermal and 31P-NMR studies to elucidate sumatriptan succinate entrapment behavior in phosphatidylcholine/cholesterol liposomes. Comparative 31P-NMR analysis on negatively and positively-charged liposomes.
Colloids Surf. B Biointerfaces 105 , 14-23, (2013) In this paper, two techniques, differential scanning calorimetry (DSC) and phosphorus nuclear magnetic resonance ((31)P-NMR), have been used to characterize sumatriptan succinate-loaded charged liposo... |
|
|
Identification of active ingredients in Wuzhuyu decoction improving migraine in mice by spectral efficiency association.
Mol. Med. Report. 12 , 1524-34, (2015) Wuzhuyu decoction is a traditional Chinese medicine used for the effective treatment of migraines, termed 'Jueyin headache', in China. However, there have been few investigations to clarify the compos... |
| Sumatriptan succinate |
| 1-{3-[2-(dimethylamino)ethyl]-1H-indol-5-yl}-N-methylmethanesulfonamide butanedioate (1:1) |
| 3-[2-(Dimethylamino)ethyl]-N-methyl-1H-indole-5-methanesulfonamide,butanedioate(1:1) |
| 1-(3-(2-(Dimethylamino)ethyl)-1H-indol-5-yl)-N-methylmethanesulfonamide succinate |
| 1H-Indole-5-methanesulfonamide, 3-[2-(dimethylamino)ethyl]-N-methyl-, butanedioate (1:1) |
| 1-{3-[2-(Dimethylamino)ethyl]-1H-indol-5-yl}-N-methylmethanesulfonamide succinate |
| 3-[2-(Dimethylamino)ethyl]-1H-indol-5-yl-N-methylmethanesulfonamide Succinate |
| acide butanedioïque - 1-{3-[2-(diméthylamino)éthyl]-1H-indol-5-yl}-N-méthylméthanesulfonamide (1:1) |
| Benzamide, 4-(aminosulfonyl)-N-[(1-ethyl-2-pyrrolidinyl)methyl]-2-methoxy- |
| MFCD00866222 |
| N-[(1-Ethyl-2-pyrrolidinyl)methyl]-2-methoxy-4-sulfamoylbenzamide |
| 1-{3-[2-(dimethylamino)ethyl]-1H-indol-5-yl}-N-methylmethanesulfonamide butanedioate |
| Butandisäure--1-{3-[2-(dimethylamino)ethyl]-1H-indol-5-yl}-N-methylmethansulfonamid(1:1) |
| Succinic acid - 1-{3-[2-(dimethylamino)ethyl]-1H-indol-5-yl}-N-methylmethanesulfonamide (1:1) |
| 1-{3-[2-(Dimethylamino)ethyl]-1H-indol-5-yl}-N-methylmethanesulfonamide succinate (1:1) |