18797-79-0
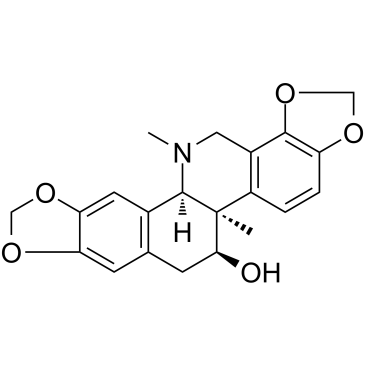
| Name | Corynoline |
|---|---|
| Synonyms |
(11S,13R,14R)-corynoline
13-Methylchelidonine 13-methyl-chelidonine Corynoline |
| Description | Corynoline, isolated from Corydalis incise (Papaveraceae), is a reversible and noncompetitive acetylcholinesterase (AChE) inhibitor with an IC50 of 30.6 μM[1]. Corynoline exhibits anti-inflammatory activity by activating Nrf2[2]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 504.2±50.0 °C at 760 mmHg |
| Melting Point | 217-218ºC |
| Molecular Formula | C21H21NO5 |
| Molecular Weight | 367.395 |
| Flash Point | 258.7±30.1 °C |
| Exact Mass | 367.141968 |
| PSA | 60.39000 |
| LogP | 3.23 |
| Vapour Pressure | 0.0±1.4 mmHg at 25°C |
| Index of Refraction | 1.659 |
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H300 + H330 |
| Precautionary Statements | P260-P284-P301 + P310 + P330-P304 + P340 + P310-P403 + P233 |
| Hazard Codes | Xi |
| Safety Phrases | 24/25 |
| RIDADR | UN 1544PSN1 6.1 / PGII |
| Precursor 0 | |
|---|---|
| DownStream 1 | |