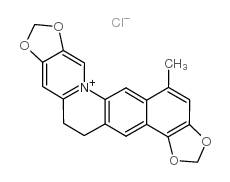
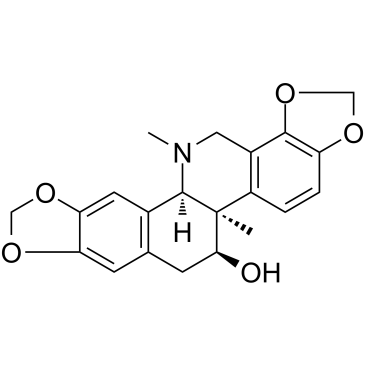
18797-80-3
| Name | Acetylcorynoline |
|---|---|
| Synonyms |
(5bR)-6t-acetoxy-5b,13-dimethyl-(5br,12bc)-5b,6,7,12b,13,14-hexahydro-[1,3]dioxolo[4,5-i][1,3]dioxolo[4',5':4,5]benzo[1,2-c]phenanthridine
corynoline acetate (+)-O-acetyl-corynoline N1513 Acetylcorynoline (+)-Corynolin-acetat O-Acetyl-13-methylchelidonine O-acetyl-13-methyl-chelidonine (+)-Corynolin-O-acetat |
| Description | Acetylcorynoline is the major alkaloid component derived from Corydalis bungeana, and has anti-inflammatory properties[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Acetylcorynoline significantly inhibits the secretion of tumor necrosis factor-α, interleukin-6, and interleukin-12p70 by LPS-stimulated Dendritic cells (DCs)[1]. Acetylcorynoline significantly inhibits LPS-induced activation of IκB kinase and mitogen-activated protein kinase[1]. |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 499.1±45.0 °C at 760 mmHg |
| Molecular Formula | C23H23NO6 |
| Molecular Weight | 409.432 |
| Flash Point | 255.7±28.7 °C |
| Exact Mass | 409.152527 |
| PSA | 66.46000 |
| LogP | 4.05 |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C |
| Index of Refraction | 1.667 |
| Storage condition | 2-8C |
|
~% 
18797-80-3 |
| Literature: Tetrahedron Letters, , vol. 29, # 50 p. 6621 - 6624 |
|
~% 
18797-80-3 |
| Literature: Tetrahedron Letters, , vol. 29, # 50 p. 6621 - 6624 |
|
~% 
18797-80-3 |
| Literature: Tetrahedron Letters, , vol. 29, # 50 p. 6621 - 6624 |
|
~% 
18797-80-3 |
| Literature: Tetrahedron Letters, , vol. 29, # 50 p. 6621 - 6624 |
|
~% 
18797-80-3 |
| Literature: Tetrahedron Letters, , vol. 29, # 50 p. 6621 - 6624 |
|
~% 
18797-80-3 |
| Literature: Tetrahedron Letters, , vol. 29, # 50 p. 6621 - 6624 |
|
~% 
18797-80-3 |
| Literature: Tetrahedron Letters, , vol. 29, # 50 p. 6621 - 6624 |
|
~% 
18797-80-3 |
| Literature: Tetrahedron Letters, , vol. 29, # 50 p. 6621 - 6624 |
| Precursor 5 | |
|---|---|
| DownStream 0 | |