UNII:3I7LZ8M32B
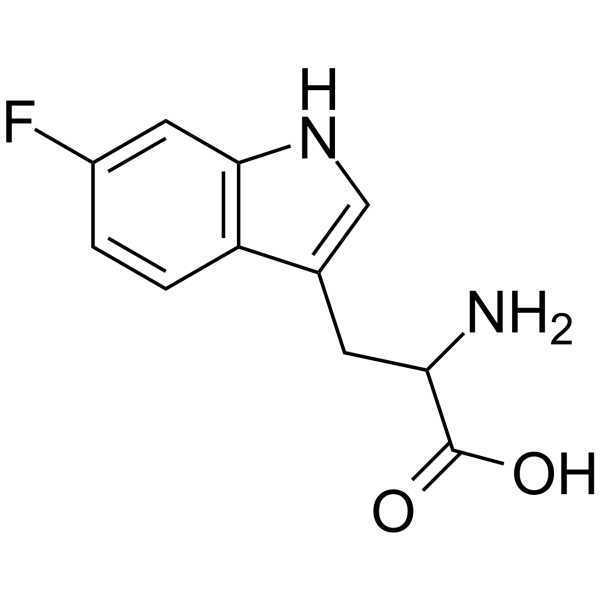
UNII:3I7LZ8M32B structure
|
Common Name | UNII:3I7LZ8M32B | ||
|---|---|---|---|---|
| CAS Number | 7730-20-3 | Molecular Weight | 222.216 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 450.7±45.0 °C at 760 mmHg | |
| Molecular Formula | C11H11FN2O2 | Melting Point | 280-285 °C (dec.)(lit.) | |
| MSDS | Chinese USA | Flash Point | 226.4±28.7 °C | |
|
Substrate promiscuity of the cyclic dipeptide prenyltransferases from Aspergillus fumigatus ( section sign).
J. Nat. Prod. 72 , 44-52, (2009) This study reports that a series of tryptophan derivatives with modifications on the side chain or at the indole ring were accepted by two cyclic dipeptide prenyltransferases, CdpNPT and FtmPT1, and converted to prenylated derivatives. The structures of the e... |
|
|
Cloning of the trp gene cluster from a tryptophan-hyperproducing strain of Corynebacterium glutamicum: identification of a mutation in the trp leader sequence.
Appl. Environ. Microbiol. 59(3) , 791-9, (1993) Corynebacterium glutamicum ATCC 21850 produces up to 5 g of extracellular L-tryptophan per liter in broth culture and displays resistance to several synthetic analogs of aromatic amino acids. Here we report the cloning of the tryptophan biosynthesis (trp) gen... |
|
|
Atomic mutations at the single tryptophan residue of human recombinant annexin V: effects on structure, stability, and activity.
Biochemistry 38(33) , 10649-59, (1999) The single tryptophan residue (Trp187) of human recombinant annexin V, containing 320 residues and 5328 atoms, was replaced with three different isosteric analogues where hydrogen atoms at positions 4, 5, and 6 in the indole ring were exchanged with fluorine.... |
|
|
Incorporation of tryptophan analogues into staphylococcal nuclease, its V66W mutant, and Delta 137-149 fragment: spectroscopic studies.
Biochemistry 37(25) , 8938-46, (1998) We have biosynthetically incorporated several tryptophan analogues into three forms of Staphylococcal nuclease to investigate the spectroscopic characteristics of these "intrinsic" probes and their effect on the structure of the proteins. The set of tryptopha... |
|
|
Phosphorescence and optically detected magnetic resonance characterization of the environments of tryptophan analogues in staphylococcal nuclease, its V66W mutant, and Delta 137-149 fragment.
Biochemistry 37(25) , 8954-64, (1998) Phosphorescence and optically detected magnetic resonance (ODMR) measurements are reported on the triplet states of the tryptophan analogues, 7-azatryptophan (7AW), 5-hydroxytryptophan (5HW), and 4-, 5-, and 6-fluorotryptophan (4FW, 5FW, 6FW), when incorporat... |
|
|
Catalytic site conformations in human PNP by 19F-NMR and crystallography.
Chem. Biol. 20(2) , 212-22, (2013) Purine nucleoside phosphorylase (PNP) is a target for leukemia, gout, and autoimmune disorders. Dynamic motion of catalytic site loops has been implicated in catalysis, but experimental evidence was lacking. We replaced catalytic site groups His257 or His64 w... |
|
|
Comparison of the folding mechanism of highly homologous proteins in the lipid-binding protein family.
Proteins 75(4) , 799-806, (2009) The folding mechanism of two closely related proteins in the intracellular lipid-binding protein family, human bile acid-binding protein (hBABP), and rat bile acid-binding protein (rBABP) were examined. These proteins are 77% identical (93% similar) in sequen... |
|
|
Auto-induction medium containing glyphosate for high-level incorporation of unusual aromatic amino acids into proteins.
Biotechniques 49 , 659-661, (2010) We describe the use of an auto-induction medium containing N-(phosphono-methyl)glycine (glyphosate) as a means for high-level introduction of nonstandard aromatic amino acids into a protein. We illustrate this approach by preparing maltose binding protein (MB... |
|
|
19F nuclear magnetic resonance studies of 6-fluorotryptophan-substituted rat cellular retinol binding protein II produced in Escherichia coli. An analysis of four tryptophan substitution mutants and their interactions with all-trans-retinol.
J. Biol. Chem. 265(20) , 11549-54, (1990) Rat cellular retinol binding protein (CRBP II) is a 134-amino acid intracellular protein synthesized in the polarized absorptive cells of the intestine. We have previously used 19F nuclear magnetic resonance (NMR) spectroscopy to survey the structural effects... |
|
|
Nuclear magnetic resonance studies of 6-fluorotryptophan-substituted rat cellular retinol-binding protein II produced in Escherichia coli. Analysis of the apoprotein and the holoprotein containing bound all-trans-retinol and all-trans-retinal.
J. Biol. Chem. 264(29) , 17041-8, (1989) Rat cellular retinol-binding protein II (CRBP II) is a 15.6-kDa intestinal protein which binds all-trans-retinol and all-trans-retinal but not all-trans-retinoic acid. We have previously analyzed the interaction of Escherichia coli-derived rat apoCRBP II with... |