15-HEPE
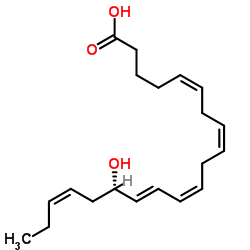
15-HEPE structure
|
Common Name | 15-HEPE | ||
|---|---|---|---|---|
| CAS Number | 86282-92-0 | Molecular Weight | 318.450 | |
| Density | 1.0±0.1 g/cm3 | Boiling Point | 488.0±45.0 °C at 760 mmHg | |
| Molecular Formula | C20H30O3 | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 263.1±25.2 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
|
Normalisation genes for expression analyses in the brown alga model Ectocarpus siliculosus.
BMC Mol. Biol. 9 , 75, (2008) Brown algae are plant multi-cellular organisms occupying most of the world coasts and are essential actors in the constitution of ecological niches at the shoreline. Ectocarpus siliculosus is an emerging model for brown algal research. Its genome has been seq... |
|
|
Mass spectrometry of underivatized 15-hydroxyeicosatetraenoic acid and 15-hydroxyeicosapentaenoic acid.
Biomed. Environ. Mass Spectrom. 17(6) , 459-61, (1988) A direct mass spectrometric method for 15-hydroxy metabolites of arachidonic acid and of eicosapentaenoic acid is described. 15-Hydroxyeicosanoids have the capacity to inhibit the formation of leukotrienes, potent mediators of inflammation formed by the 5-lip... |
|
|
[Endogenous ligands for PPARs].
Nihon Rinsho. 63(4) , 578-83, (2005) Peroxisome proliferator-activated receptors (PPARs), a family of three nuclear receptors/transcription factors, are widely recognized as molecular targets for drugs against lifestyle related disease. In spite of intensive search for natural ligands, no truly ... |
|
|
Novel functional sets of lipid-derived mediators with antiinflammatory actions generated from omega-3 fatty acids via cyclooxygenase 2-nonsteroidal antiinflammatory drugs and transcellular processing.
J. Exp. Med. 192(8) , 1197-204, (2000) Aspirin therapy inhibits prostaglandin biosynthesis without directly acting on lipoxygenases, yet via acetylation of cyclooxygenase 2 (COX-2) it leads to bioactive lipoxins (LXs) epimeric at carbon 15 (15-epi-LX, also termed aspirin-triggered LX [ATL]). Here,... |
|
|
Involvement of 15-lipoxygenase in the inflammatory arthritis.
J. Cell. Biochem. 113(7) , 2279-89, (2012) 15-Lipoxygenase (15-LOX) is involved in many pathological processes. The aim of this study is to examine the role of 15-LOX in the matrix metalloproteinase (MMP) expression and inflammatory arthritis. It was found that treatment of 15-LOX downstream product o... |
|
|
15-Hydroperoxyeicosapentaenoic acid inhibits arachidonic acid metabolism in rabbit platelets more potently than eicosapentaenoic acid.
Biochim. Biophys. Acta 1300(3) , 171-6, (1996) The effect of 15-hydroperoxy-5,8,11,13,15-eicosapentaenoic acid (15-HPEPE), a hydroperoxy adduct of eicosapentaenoic acid (EPA), on the formation of 12-hydroxy-5,8,10,14-eicosatetraenoic acid (12-HETE), thromboxane (TX) B2 and 12-hydroxy-5,8,10-heptadecatrien... |
|
|
ALOX5 gene variants affect eicosanoid production and response to fish oil supplementation.
J. Lipid Res. 52(5) , 991-1003, (2011) The objective of this study was to determine whether 5-lipoxygenase (ALOX5) gene variants associated with cardiovascular disease affect eicosanoid production by monocytes. The study was a randomized, double-masked, parallel intervention trial with fish oil (5... |
|
|
Overexpression of 15-lipoxygenase-1 induces growth arrest through phosphorylation of p53 in human colorectal cancer cells.
Mol. Cancer Res. 3(9) , 511-7, (2005) To investigate the function of 15-lipoxygenase-1 (15-LOX-1) in human colorectal cancer, we overexpressed 15-LOX-1 in HCT-116 human colorectal cancer cells. Clones expressing the highest levels of 15-LOX-1 displayed reduced viability compared with the HCT-116-... |
|
|
Probing the skin permeation of eicosapentaenoic acid and ketoprofen 2. Comparative depth profiling and metabolism of eicosapentaenoic acid.
Eur. J. Pharm. Biopharm. 67(1) , 156-65, (2007) Unexpected enhancement of the topical delivery of eicosapentaenoic acid (EPA) across porcine skin was observed previously when fish oil was co-formulated with ketoprofen. In the current work depth profile analysis was used to probe the epidermal conversion of... |
|
|
Effects of eicosapentaenoic acid and its 15-hydroperoxy and 15-hydroxy derivatives on glucosamine synthetase activity in rabbit gastric mucosa.
Biochem. Mol. Biol. Int. 46(1) , 157-63, (1998) The effects of eicosapentaenoic acid and its 15-hydroperoxy and 15-hydroxy adducts on the activity of glucosamine synthetase, the rate-limiting enzyme of mucus synthesis, in rabbit gastric antral mucosa were examined. 15-Hydroperoxy-eicosapentaenoic acid inhi... |