Methyl (9E)-9-octadecenoate
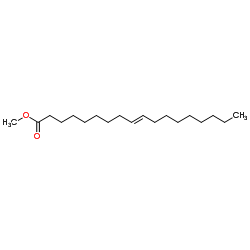
Methyl (9E)-9-octadecenoate structure
|
Common Name | Methyl (9E)-9-octadecenoate | ||
|---|---|---|---|---|
| CAS Number | 1937-62-8 | Molecular Weight | 296.488 | |
| Density | 0.9±0.1 g/cm3 | Boiling Point | 351.4±0.0 °C at 760 mmHg | |
| Molecular Formula | C19H36O2 | Melting Point | 41892ºC | |
| MSDS | Chinese USA | Flash Point | 92.4±20.4 °C | |
| Symbol |




GHS02, GHS07, GHS08, GHS09 |
Signal Word | Danger | |
|
Optimization of supercritical fluid consecutive extractions of fatty acids and polyphenols from Vitis vinifera grape wastes.
J. Food Sci. 80(1) , E101-7, (2015) In this study, supercritical fluid extraction has been successfully applied to a sequential fractionation of fatty acids and polyphenols from wine wastes (2 different vitis vinifera grapes). To this aim, in a 1st step just fatty acids were extracted and in a ... |
|
|
Effect of different biosynthetic precursors on the production of nargenicin A1 from metabolically engineered Nocardia sp. CS682.
J. Microbiol. Biotechnol. 22(8) , 1127-32, (2012) Nargenicin A1 is a 28-membered polyketide macrolide, with antibacterial activity against methicillin-resistant Staphylococcus aureus, produced by Nocardia sp. CS682. In this study, the production of nargenicin A1 was improved by enhancing the supply of differ... |
|
|
Quantitative analysis of pheromone-binding protein specificity.
Insect Mol. Biol. 22(1) , 31-40, (2013) Many pheromones have very low water solubility, posing experimental difficulties for quantitative binding measurements. A new method is presented for determining thermodynamically valid dissociation constants for ligands binding to pheromone-binding proteins,... |
|
|
Rate constants of oxidation of unsaturated fatty esters studied by chemiluminescence
Chem. Phys. Lipids 165(7) , 753-9, (2012) Graphical abstract |
|
|
A detailed identification study on high-temperature degradation products of oleic and linoleic acid methyl esters by GC-MS and GC-FTIR.
Chem. Phys. Lipids 165(3) , 338-47, (2012) GC-MS and GC-FTIR were complementarily applied to identify oxidation compounds formed under frying conditions in methyl oleate and linoleate heated at 180°C. The study was focused on the compounds that originated through hydroperoxide scission that remain att... |
|
|
Catalytic epoxidation of a technical mixture of methyl oleate and methyl linoleate in ionic liquids using MoO(O2)2.2QOH (QOH = 8-quinilinol) as catalyst and NaHCO3 as co-catalyst.
Molecules 14(8) , 2935-46, (2009) The oxo-diperoxo molybdenum(VI) complex MoO(O(2))(2).2QOH (QOH = 8-quinilinol) was prepared and characterized by elemental analysis, IR and UV-Vis spectra. The ionic liquids (ILs) [bmim][BF(4)], [hydemim][BF(4)], and [bmim][PF(6)] were characterized by (1)H-N... |
|
|
Preparation of acetonides from soybean oil, methyl soyate, and fatty esters.
J. Agric. Food Chem. 59(7) , 3066-70, (2011) This paper describes the preparation of a new type of branched vegetable oil and its methyl ester that involves the formation of acetonides. A facile and environmentally friendly synthesis has been found to produce acetonides that entails the use of ferric ch... |
|
|
[Autooxidation of a mixture of lemon essential oils, methyl linolenoate, and methyl oleinate].
Prikl. Biokhim. Mikrobiol. 46(5) , 599-604, (2010) Stability of components of a mixture of methyl linolenoate and methyl oleinate with two lemon (Citrus limon L.) essential oils in hexane during their autooxidation in light was studied by gas chromatography. The essential oils differed by their quantitative r... |
|
|
High-level production of extracellular lipase by Yarrowia lipolytica mutants from methyl oleate.
New Biotechnology 28(6) , 756-60, (2011) The yeast Yarrowia lipolytica degrades efficiently low-cost hydrophobic substrates for the production of various added-value products such as lipases. To obtain yeast strains producing high levels of extracellular lipase, Y. lipolytica DSM3286 was subjected t... |
|
|
A novel biocompatible adhesive incorporating plant-derived monomers.
J. Biomed. Mater. Res. A 91(2) , 378-84, (2009) We describe a new class of biomaterials with potential for a variety of applications in tissue engineering, wound healing, and transdermal drug delivery. These materials are based on oleic methyl ester (OME), which is derived from various plant oils including... |