Drospirenone
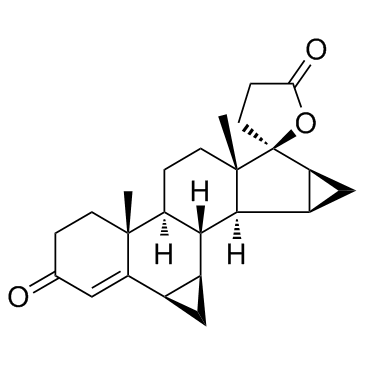
Drospirenone structure
|
Common Name | Drospirenone | ||
|---|---|---|---|---|
| CAS Number | 67392-87-4 | Molecular Weight | 366.493 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 552.2±50.0 °C at 760 mmHg | |
| Molecular Formula | C24H30O3 | Melting Point | 196-200ºC | |
| MSDS | Chinese USA | Flash Point | 241.6±30.2 °C | |
| Symbol |

GHS08 |
Signal Word | Danger | |
|
Postural tachycardia syndrome (POTS).
Circulation 127(23) , 2336-42, (2013)
|
|
|
Sexual function, mood and menopause symptoms in Lithuanian postmenopausal women.
Climacteric 16(1) , 185-93, (2013) To assess sexual function in a clinical sample of Lithuanian postmenopausal women and identify the most important determinants of sexual function, including the use of hormone replacement therapy (HT), emotional status and menopausal symptoms.Three hundred po... |
|
|
Liquid chromatography with diode array detection and multivariate curve resolution for the selective and sensitive quantification of estrogens in natural waters.
Anal. Chim. Acta 835 , 19-28, (2014) Following the green analytical chemistry principles, an efficient strategy involving second-order data provided by liquid chromatography (LC) with diode array detection (DAD) was applied for the simultaneous determination of estriol, 17β-estradiol, 17α-ethiny... |
|
|
In search of the best combined oral contraceptive for treatment of polycystic ovary syndrome.
Fertil. Steril. 98(4) , 825-6, (2012)
|
|
|
Comparative study of the therapeutic effects of oral contraceptive pills containing desogestrel, cyproterone acetate, and drospirenone in patients with polycystic ovary syndrome.
Fertil. Steril. 98(4) , 1053-9, (2012) To compare the effects of oral contraceptive pills containing desogestrel, cyproterone acetate, and drospirenone, in polycystic ovary syndrome (PCOS), after 6 and 12 months of therapy.Double-blind randomized controlled trial.Gynecologic clinic of the first au... |
|
|
Comparison of two oral contraceptive forms containing cyproterone acetate and drospirenone in the treatment of patients with polycystic ovary syndrome: a randomized clinical trial.
Arch. Gynecol. Obstet. 290(2) , 321-8, (2014) To compare the effects of combined oral contraceptives (OCs) containing cyproterone acetate and drospirenone in the treatment of polycystic ovary syndrome (PCOS).Fifty-two patients with PCOS were randomized in two groups: group A (n = 26) received 0.035 mg et... |
|
|
The “newer” progestogens and postmenopausal hormone therapy (HRT)
J. Steroid Biochem. Mol. Biol. 142 , 48-51, (2014) • The “newer” progestogens have a high bioavailability. • The “newer” progestogens have a sufficient progestogenic effect. • The “newer” progestogens do not cause undue side effects. • The “newer” progestogens do not jeopardize lipid- and carbohydrate metabol... |
|
|
Low-dose estrogen and drospirenone combination: effects on metabolism and endothelial function in postmenopausal women with metabolic syndrome.
Clin. Exp. Obstet. Gynecol. 40(2) , 233-5, (2013) Metabolic syndrome and endothelial dysfunction play a relevant role in the cardiovascular risk in post-menopause. The aim of the study was to assess the effects of a low-dose hemihydrate estradiol and drospirenone combination on cardiovascular risk parameters... |
|
|
Dual suppression with oral contraceptive pills in GnRH antagonist cycles for patients with polycystic ovary syndrome undergoing intracytoplasmic sperm injection.
Eur. J. Obstet. Gynecol. Reprod. Biol. 183 , 137-40, (2014) To evaluate the effects of a gonadotropin-releasing hormone (GnRH) antagonist protocol, with or without oral contraceptive pill (OCP) pretreatment, in patients with polycystic ovary syndrome (PCOS) undergoing intracytoplasmic sperm injection (ICSI).In this re... |
|
|
Progestin-dependent effect of oral contraceptives on plasma aldosterone/renin ratio.
Clin. Biochem. 45(16-17) , 1516-8, (2012) Estrogens in oral contraceptives (OC) may influence plasma aldosterone/plasma renin activity (ALD/PRA) and plasma aldosterone/plasma renin concentration (ALD/DRC) ratios, but the effect of progestins on these ratios has not been sufficiently studied so far.PR... |