Guaiacol
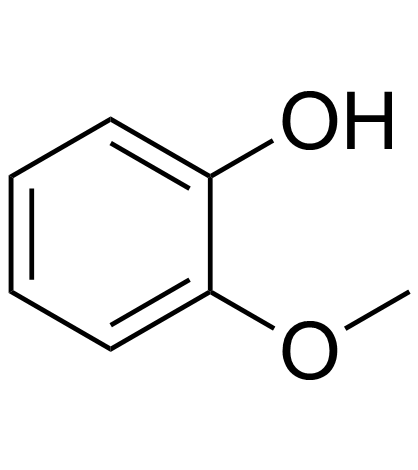
Guaiacol structure
|
Common Name | Guaiacol | ||
|---|---|---|---|---|
| CAS Number | 90-05-1 | Molecular Weight | 124.14 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 207.5±29.0 °C at 760 mmHg | |
| Molecular Formula | C7H8O2 | Melting Point | 26-29 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 79.3±24.3 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of GuaiacolGuaiacol, a phenolic compound isolated from Guaiac resin, inhibits LPS-stimulated COX-2 expression and NF-κB activation[1]. Anti-inflammatory activity[1]. |
| Name | guaiacol |
|---|---|
| Synonym | More Synonyms |
| Description | Guaiacol, a phenolic compound isolated from Guaiac resin, inhibits LPS-stimulated COX-2 expression and NF-κB activation[1]. Anti-inflammatory activity[1]. |
|---|---|
| Related Catalog | |
| Target |
COX-2 NF-κB |
| In Vitro | Guaiacol inhibits LPS-stimulated nuclear factor kappa B (NF-κB) activation and cyclooxygenase (COX)-2 gene expression in cells of the RAW 264.7 murine macrophage cell line. Phenolic compounds such as Phenol, Eugenol, Guaiacol and Vanillin inhibit sheep vesicular gland prostaglandin cyclooxygenase, as indicated by their 50% inhibition concentrations, which decline in the following order: Phenol (1600 μM) > Vanillin (>500 μM) > Guaiacol (72 μM) > Eugenol (12 μM)[1]. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 207.5±29.0 °C at 760 mmHg |
| Melting Point | 26-29 °C(lit.) |
| Molecular Formula | C7H8O2 |
| Molecular Weight | 124.14 |
| Flash Point | 79.3±24.3 °C |
| PSA | 29.46000 |
| LogP | 2.11 |
| Vapour density | 4.27 (vs air) |
| Vapour Pressure | 0.2±0.4 mmHg at 25°C |
| Index of Refraction | 1.538 |
| InChIKey | LHGVFZTZFXWLCP-UHFFFAOYSA-N |
| SMILES | COc1ccccc1O |
| Storage condition | Refrigerator |
| Stability | Stable, but air and light sensitive. Combustible. Incompatible with strong oxidizing agents. |
| Water Solubility | 17 g/L (15 ºC) |
| Freezing Point | 28℃ |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H315-H319 |
| Precautionary Statements | P280-P301 + P312 + P330-P305 + P351 + P338-P337 + P313 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Faceshields;Gloves |
| Hazard Codes | Xn:Harmful |
| Risk Phrases | R22;R36/38 |
| Safety Phrases | S26 |
| RIDADR | 2810 |
| WGK Germany | 1 |
| RTECS | SL7525000 |
| Packaging Group | II |
| Hazard Class | 6.1(b) |
| HS Code | 2909500000 |
| Precursor 10 | |
|---|---|
| DownStream 10 | |
| HS Code | 2909500000 |
|---|---|
| Summary | 2909500000 ether-phenols, ether-alcohol-phenols and their halogenated, sulphonated, nitrated or nitrosated derivatives VAT:17.0% Tax rebate rate:9.0% Supervision conditions:none MFN tariff:5.5% General tariff:30.0% |
|
Oxidative stress influences positive strand RNA virus genome synthesis and capping.
Virology 475 , 219-29, (2014) Flaviviruses are 5' capped positive-stranded RNA viruses that replicate their genomes within endoplasmic reticulum-derived vesicles. Flaviviruses are well known to induce oxidative stress late in infe... |
|
|
Fungal metabolite nigerloxin ameliorates diabetic nephropathy and gentamicin-induced renal oxidative stress in experimental rats.
Naunyn Schmiedebergs Arch. Pharmacol. 387(9) , 849-59, (2014) Elevated polyol pathway enzyme activities and oxidative stress play an important role in the development and progression of diabetic nephropathy. Here, we investigated the beneficial influence of nige... |
|
|
Lignin Modification for Biopolymer/Conjugated Polymer Hybrids as Renewable Energy Storage Materials.
ChemSusChem 8 , 4081-5, (2015) Lignin derivatives, which arise as waste products from the pulp and paper industry and are mainly used for heating, can be used as charge storage materials. The charge storage function is a result of ... |
| 5-fluoro-2,3-dihydro-1h-isoindole |
| 2-Methoxyphenol |
| methoxyphenol |
| 2,3-dihydro-6-fluoro-1H-indole |
| o-Methoxyphenol |
| Guaiacol |
| MFCD00002185 |
| Indoline, 6-fluoro- |
| 2-Hydroxyanisole |
| 2-Methoxy-phenol |
| o-Methylcatechol |
| 2,3-dihydro-6-fluoroindole |
| Catechol, Methyl |
| o-methoxy-Phenol |
| 6-Fluor-indolin |
| 6-Fluoroindoline |
| ORTHO-METHOXYPHENOL |
| 6-fluoro-2,3-dihydro-indole |
| Phenol, 2-methoxy- |
| o-methoxy catechol |
| O-Methyl catechol |
| 1H-Indole, 6-fluoro-2,3-dihydro- |
| Indoline,6-fluoro |
| Methylcatechol |
| EINECS 201-964-7 |
| Catechol monomethyl ether |
| o-Hydroxyanisole |
 CAS#:5720-06-9
CAS#:5720-06-9 CAS#:578-57-4
CAS#:578-57-4 CAS#:529-28-2
CAS#:529-28-2 CAS#:766-51-8
CAS#:766-51-8 CAS#:10535-17-8
CAS#:10535-17-8 CAS#:121-33-5
CAS#:121-33-5![1,2-dimethoxy-4-[(2-methoxyphenoxy)methyl]benzene Structure](https://image.chemsrc.com/caspic/428/10548-82-0.png) CAS#:10548-82-0
CAS#:10548-82-0 CAS#:100-66-3
CAS#:100-66-3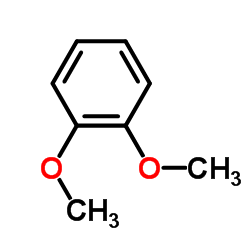 CAS#:91-16-7
CAS#:91-16-7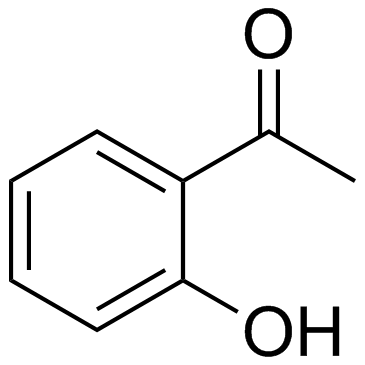 CAS#:118-93-4
CAS#:118-93-4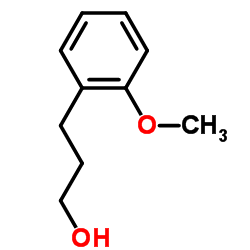 CAS#:10493-37-5
CAS#:10493-37-5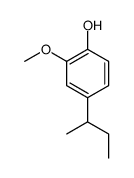 CAS#:110030-28-9
CAS#:110030-28-9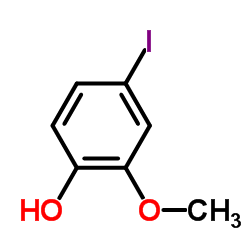 CAS#:203861-62-5
CAS#:203861-62-5 CAS#:111726-46-6
CAS#:111726-46-6![1-[2-(2-METHOXYPHENOXY)ETHYL]-PIPERIDINE structure](https://image.chemsrc.com/caspic/349/105602-16-2.png) CAS#:105602-16-2
CAS#:105602-16-2![3-[(2-methoxyphenoxy)methyl]-4-phenyl-1H-1,2,4-triazole-5-thione structure](https://image.chemsrc.com/caspic/031/107952-04-5.png) CAS#:107952-04-5
CAS#:107952-04-5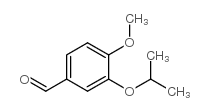 CAS#:34123-66-5
CAS#:34123-66-5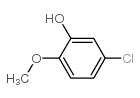 CAS#:3743-23-5
CAS#:3743-23-5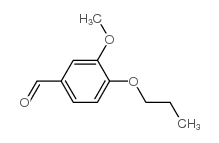 CAS#:57695-98-4
CAS#:57695-98-4
