2-Methyl-2-propanyl (2-oxoethyl)carbamate
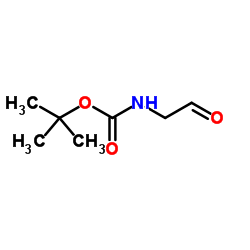
2-Methyl-2-propanyl (2-oxoethyl)carbamate structure
|
Common Name | 2-Methyl-2-propanyl (2-oxoethyl)carbamate | ||
|---|---|---|---|---|
| CAS Number | 89711-08-0 | Molecular Weight | 159.183 | |
| Density | 1.0±0.1 g/cm3 | Boiling Point | 237.2±23.0 °C at 760 mmHg | |
| Molecular Formula | C7H13NO3 | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 97.3±22.6 °C | |
| Name | tert-butyl N-(2-oxoethyl)carbamate |
|---|---|
| Synonym | More Synonyms |
| Density | 1.0±0.1 g/cm3 |
|---|---|
| Boiling Point | 237.2±23.0 °C at 760 mmHg |
| Molecular Formula | C7H13NO3 |
| Molecular Weight | 159.183 |
| Flash Point | 97.3±22.6 °C |
| Exact Mass | 159.089539 |
| PSA | 55.40000 |
| LogP | 0.88 |
| Vapour Pressure | 0.0±0.5 mmHg at 25°C |
| Index of Refraction | 1.434 |
| InChIKey | ACNRTYKOPZDRCO-UHFFFAOYSA-N |
| SMILES | CC(C)(C)OC(=O)NCC=O |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |
| Precursor 10 | |
|---|---|
| DownStream 3 | |
|
Synthesis of fused heteroarylprolines and pyrrolopyrroles.
J. Org. Chem. 69 , 4656-4662, (2004) Fused heteroarylprolines were prepared starting from 4-oxo-N-(PhF)proline benzyl ester (6, PhF = 9-(9-phenylfluorenyl)) following two approaches. First, allylation of oxoproline 6 followed by Wacker o... |
|
|
Tumor-targeting of EGFR inhibitors by hypoxia-mediated activation.
Angew. Chem. Int. Ed. Engl. 53(47) , 12930-5, (2014) The development of receptor tyrosine-kinase inhibitors (TKIs) was a major step forward in cancer treatment. However, the therapy with TKIs is limited by strong side effects and drug resistance. The ai... |
|
|
Single cell imaging of Bruton's tyrosine kinase using an irreversible inhibitor.
Sci. Rep. 4 , 4782, (2014) A number of Bruton's tyrosine kinase (BTK) inhibitors are currently in development, yet it has been difficult to visualize BTK expression and pharmacological inhibition in vivo in real time. We synthe... |
| Carbamic acid, N-(2-oxoethyl)-, 1,1-dimethylethyl ester |
| t-butyl n-(2-oxoethyl)cabamate |
| N-tert-butoxycarbonylglycinal |
| tert-butyl 2-oxoethylcarbamate |
| 2-(t-butoxycarbonylamino)acetaldehyde |
| N-(tert-butoxycarbonyl)-2-amino-acetaldehyde |
| (2-oxo-ethyl)-carbamic acid-tert-butylester |
| 2-Methyl-2-propanyl (2-oxoethyl)carbamate |
| N-Boc-2-aminoacetaldehyde |
| N-(tert-Butoxycarbonyl)aminoacetaldehyde |
| boc-l-glycinal |
| MFCD01321273 |
| N-tert-butyloxycarbonyl-2-aminoacetaldehyde |
| (tert-butoxy)-N-(2-oxoethyl)carboxamide |
 CAS#:121505-93-9
CAS#:121505-93-9 CAS#:26690-80-2
CAS#:26690-80-2 CAS#:137618-48-5
CAS#:137618-48-5 CAS#:31954-27-5
CAS#:31954-27-5 CAS#:78888-18-3
CAS#:78888-18-3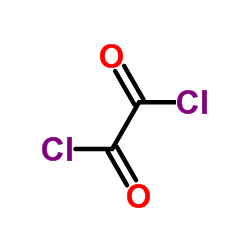 CAS#:79-37-8
CAS#:79-37-8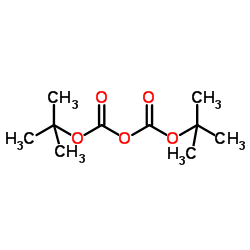 CAS#:24424-99-5
CAS#:24424-99-5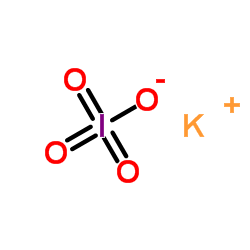 CAS#:7790-21-8
CAS#:7790-21-8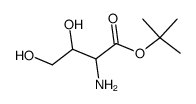 CAS#:1378293-39-0
CAS#:1378293-39-0 CAS#:616-30-8
CAS#:616-30-8 CAS#:92136-39-5
CAS#:92136-39-5![2-Butenoicacid,4-[[(1,1-dimethylethoxy)carbonyl]amino]-,(2E)-(9CI) structure](https://image.chemsrc.com/caspic/026/180913-22-8.png) CAS#:180913-22-8
CAS#:180913-22-8 CAS#:113525-94-3
CAS#:113525-94-3