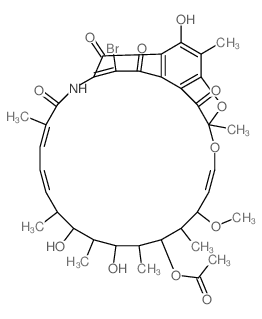
Rifaximin

Rifaximin structure
|
Common Name | Rifaximin | ||
|---|---|---|---|---|
| CAS Number | 80621-81-4 | Molecular Weight | 785.878 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | N/A | |
| Molecular Formula | C43H51N3O11 | Melting Point | 200-205ºC(dec) | |
| MSDS | Chinese USA | Flash Point | N/A | |
Use of RifaximinRifaximin(Xifaxan) is an orally administered, semi-synthetic, nonsystemic antibiotic derived from rifamycin SV with antibacterial activity.IC50 Value:Target: RNA polymerase; antibacterialRifaximin is a semisynthetic, rifamycin-based non-systemic antibiotic, meaning that very little of the drug will pass the gastrointestinal wall into the circulation as is common for other types of orally administered antibiotics. It is used in the treatment of traveler's diarrhea and hepatic encephalopathy, for which it received orphan drug status from the U.S. Food and Drug Administration in 1998. Rifaximin interferes with transcription by binding to the β-subunit of bacterial RNA polymerase. This results in the blockage of the translocation step that normally follows the formation of the first phosphodiester bond, which occurs in the transcription process. From Wikipedia. |
| Name | Rifaximin |
|---|---|
| Synonym | More Synonyms |
| Description | Rifaximin(Xifaxan) is an orally administered, semi-synthetic, nonsystemic antibiotic derived from rifamycin SV with antibacterial activity.IC50 Value:Target: RNA polymerase; antibacterialRifaximin is a semisynthetic, rifamycin-based non-systemic antibiotic, meaning that very little of the drug will pass the gastrointestinal wall into the circulation as is common for other types of orally administered antibiotics. It is used in the treatment of traveler's diarrhea and hepatic encephalopathy, for which it received orphan drug status from the U.S. Food and Drug Administration in 1998. Rifaximin interferes with transcription by binding to the β-subunit of bacterial RNA polymerase. This results in the blockage of the translocation step that normally follows the formation of the first phosphodiester bond, which occurs in the transcription process. From Wikipedia. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Melting Point | 200-205ºC(dec) |
| Molecular Formula | C43H51N3O11 |
| Molecular Weight | 785.878 |
| Exact Mass | 785.352356 |
| PSA | 198.38000 |
| LogP | 3.22 |
| Index of Refraction | 1.634 |
| InChIKey | NZCRJKRKKOLAOJ-KOPWYESNSA-N |
| SMILES | COC1C=COC2(C)Oc3c(C)c(O)c4c(O)c(c5c(nc6cc(C)ccn65)c4c3C2=O)NC(=O)C(C)=CC=CC(C)C(O)C(C)C(O)C(C)C(OC(C)=O)C1C |
| Storage condition | 0-6°C |
| Water Solubility | ethanol: soluble1mg/mL |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |
| WGK Germany | 2.0 |
| RTECS | KD1576000 |
|
~% 
Rifaximin CAS#:80621-81-4 |
| Literature: Journal of Medicinal Chemistry, , vol. 28, # 7 p. 960 - 963 |
|
~% 
Rifaximin CAS#:80621-81-4 |
| Literature: Journal of Medicinal Chemistry, , vol. 28, # 7 p. 960 - 963 |
| Precursor 2 | |
|---|---|
| DownStream 0 | |
|
Methane biogenesis during sodium azide-induced chemical hypoxia in rats.
Am. J. Physiol. Cell Physiol. 304(2) , C207-14, (2013) Previous studies demonstrated methane generation in aerobic cells. Our aims were to investigate the methanogenic features of sodium azide (NaN(3))-induced chemical hypoxia in the whole animal and to s... |
|
|
Effectiveness of different therapeutic strategies in preventing diverticulitis recurrence.
Eur. Rev. Med. Pharmacol. Sci. 17(3) , 342-8, (2013) Colonic diverticulitis shows a high recurrence rate.To assess the efficacy of three different therapeutic strategies in preventing diverticulitis recurrence.One hundred thirty patients suffering from ... |
|
|
Combination therapy for the treatment and prevention of hepatic encephalopathy.
Ann. Pharmacother. 46(11) , 1559-63, (2012) To evaluate the efficacy and safety of combination therapy for the treatment and prevention of hepatic encephalopathy (HE).A PubMed MEDLINE search was conducted (1947-June 2012) using the key terms la... |
| Normix |
| acétate de (7S,9E,11S,12R,13S,14R,15R,16R,17S,18S,19E,21Z)-2,15,17,36-tétrahydroxy-11-méthoxy-3,7,12,14,16,18,22,30-octaméthyl-6,23-dioxo-8,37-dioxa-24,27,33-triazahexacyclo[23.10.1.1.0.0.0]heptatriaconta-1(35),1(36),2,4,9,19,21,25,28,30,32,34-dodécaén-13-yle |
| rifaxidin |
| Xifaxan |
| (7S,9E,11S,12R,13S,14R,15R,16R,17S,18S,19E,21Z)-2,15,17,36-tetrahydroxy-11-methoxy-3,7,12,14,16,18,22,30-octamethyl-6,23-dioxo-8,37-dioxa-24,27,33-triazahexacyclo[23.10.1.1.0.0.0]heptatriaconta-1(35),1(36),2,4,9,19,21,25,28,30,32,34-dodecaen-13-yl acetate |
| RIFAMIXIN |
| Rifacol |
| 2,7-(Epoxy[1,11,13]pentadecatrienoimino)furo[2'',3'':7',8']naphth[1',2':4,5]imidazo[1,2-a]pyridine-1,15(2H)-dione, 25-(acetyloxy)-5,6,21,23-tetrahydroxy-27-methoxy-2,4,11,16,20,22,24,26-octamethyl-, (2S,16Z,18E,20S,21S,22R,23R,24R,25S,26R,27S,28E)- |
| l105sv |
| Rifaximin |
| l105 |
| MFCD00864973 |
| (7S,9E,11S,12R,13S,14R,15R,16R,17S,18S,19E,21Z)-2,15,17,36-Tetrahydroxy-11-methoxy-3,7,12,14,16,18,22,30-octamethyl-6,23-dioxo-8,37-dioxa-24,27,33-triazahexacyclo[23.10.1.1.0.0.0]heptatriaconta-1(35),2,4,9,19,21,25(36),26(34),28,30,32-undecaen-13-yl acetate |
| fatroximin |
| 28e))-27r |