| Description |
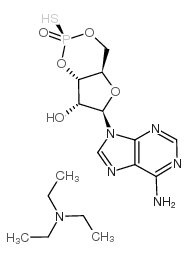
Rp-cAMPS, a cAMP analog, is a potent, competitive cAMP-induced activation of cAMP-dependent PKA I and II (Kis of 12.5 µM and 4.5 µM, respectively) antagonist. Rp-cAMPS is resistant to hydrolysis by phosphodiesterases[1][2][3][4][5][6].
|
| Related Catalog |
|
| Target |
Ki: 6.05 µM (PKA I) and 9.75 µM (PKA II)[1]
|
| In Vitro |
A membrane-permeable competitive cAMP antagonist (Rp-cAMPS) that blocks PKA activation by binding to the regulatory subunits without dissociating the kinase holoenzyme also inhibits synaptic plasticity but has no effect on normal synaptic transmission[2].
|
| In Vivo |
Rp-cAMPS (10 μM, 15 min) decreases the monosynaptic EPSCs evoked at the PB-CeLC and BLA-CeLC synapses in slices from arthritic rats but not in control neurons from normal animals. The inhibitory effect of Rp-cAMPS is significant compared to predrug (ACSF) control values obtained in the same neurons[2].
|
| References |
[1]. R J de Wit, et al. Inhibitory action of certain cyclophosphate derivatives of cAMP on cAMP-dependent protein kinases. Eur J Biochem. 1984 Jul 16;142(2):255-60. [2]. Rothermel JD, et al. A mechanistic and kinetic analysis of the interactions of the diastereoisomers of adenosine 3',5'-(cyclic)phosphorothioate with purified cyclic AMP-dependent protein kinase. Biochem J. 1988 May 1;251(3):757-62. [3]. Fu Y, et al. PKA and ERK, but not PKC, in the amygdala contribute to pain-related synaptic plasticity and behavior. Mol Pain. 2008 Jul 16;4:26. [4]. Kuriyama S, et al. Isoproterenol inhibits rod outer segment phagocytosis by both cAMP-dependent and independent pathways. Invest Ophthalmol Vis Sci. 1995 Mar;36(3):730-6. [5]. Dostmann WR, et al. Probing the cyclic nucleotide binding sites of cAMP-dependent protein kinases I and II with analogs of adenosine 3',5'-cyclic phosphorothioates. J Biol Chem. 1990 Jun 25;265(18):10484-91. [6]. Van Haastert PJ, et al. Competitive cAMP antagonists for cAMP-receptor proteins. J Biol Chem. 1984 Aug 25;259(16):10020-4.
|