Urapidil Hydrochloride
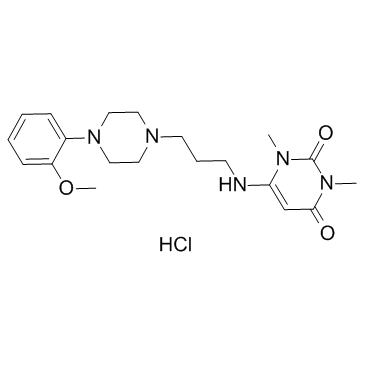
Urapidil Hydrochloride structure
|
Common Name | Urapidil Hydrochloride | ||
|---|---|---|---|---|
| CAS Number | 64887-14-5 | Molecular Weight | 423.937 | |
| Density | N/A | Boiling Point | 549ºC at 760 mmHg | |
| Molecular Formula | C20H30ClN5O3 | Melting Point | 156-158ºC | |
| MSDS | Chinese USA | Flash Point | 285.8ºC | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of Urapidil HydrochlorideUrapidil HCl is an α1-adrenoceptor antagonist and 5-HT1A receptor agonist.Target: α1-adrenoceptor; 5-HT1A receptorUrapidil hydrochloride is a hydrochloride salt form of urapidil which is α1-adrenoceptor antagonist and 5-HT1A receptor agonist with pIC50 of 6.13 and 6.4 respectively. Urapidil has an alpha-blocking effect but, unlike other alpha-blockers, also has a central sympatholytic effect mediated via stimulation of serotonin 5HT1A receptors in the central nervous system [1]. Urapidil has an alpha-blocking effect but, unlike other alpha-blockers, also has a central sympatholytic effect mediated via stimulation of serotonin 5HT1A receptors in the central nervous system. Several studies have suggested that oral urapidil is effective and well tolerated when used as second-line therapy in patients with BP inadequately controlled with other agents. Urapidil has also been shown to improve glucose and lipid metabolism in hypertensive patients with concomitant diabetes and/or hyperlipidemia [2]. |
| Name | Urapidil Hydrochloride |
|---|---|
| Synonym | More Synonyms |
| Description | Urapidil HCl is an α1-adrenoceptor antagonist and 5-HT1A receptor agonist.Target: α1-adrenoceptor; 5-HT1A receptorUrapidil hydrochloride is a hydrochloride salt form of urapidil which is α1-adrenoceptor antagonist and 5-HT1A receptor agonist with pIC50 of 6.13 and 6.4 respectively. Urapidil has an alpha-blocking effect but, unlike other alpha-blockers, also has a central sympatholytic effect mediated via stimulation of serotonin 5HT1A receptors in the central nervous system [1]. Urapidil has an alpha-blocking effect but, unlike other alpha-blockers, also has a central sympatholytic effect mediated via stimulation of serotonin 5HT1A receptors in the central nervous system. Several studies have suggested that oral urapidil is effective and well tolerated when used as second-line therapy in patients with BP inadequately controlled with other agents. Urapidil has also been shown to improve glucose and lipid metabolism in hypertensive patients with concomitant diabetes and/or hyperlipidemia [2]. |
|---|---|
| Related Catalog | |
| References |
| Boiling Point | 549ºC at 760 mmHg |
|---|---|
| Melting Point | 156-158ºC |
| Molecular Formula | C20H30ClN5O3 |
| Molecular Weight | 423.937 |
| Flash Point | 285.8ºC |
| Exact Mass | 423.203705 |
| PSA | 71.74000 |
| LogP | 1.59470 |
| Storage condition | Desiccate at RT |
| Water Solubility | H2O: soluble |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn:Harmful; |
| Risk Phrases | R22 |
| Safety Phrases | S36 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | YQ9862000 |
| HS Code | 3004909090 |
| HS Code | 3004909090 |
|---|
|
Facilitation of expulsion of ureteral stones by addition of α1-blockers to conservative therapy.
Scand. J. Urol. Nephrol. 44(6) , 420-4, (2010) An antispasmodic agent and a medicine that facilitates stone expulsion are given commonly as conservative therapy for ureteral stones in Japan. The goal of this study was to compare the efficacy of th... |
|
|
Capillary electrophoresis with end-column electrochemiluminescence for ultrasensitive determination of urapidil hydrochloride in rat plasma and its application to pharmacokinetics study.
J. Chromatogr. B. Analyt. Technol. Biomed. Life Sci. 1006 , 146-50, (2015) A simple, sensitive and selective method for determination of urapidil hydrochloride was developed using capillary electrophoresis with electrochemiluminescence (CE-ECL) technique for the first time. ... |
|
|
[Hypertensive crisis in cardiology: place of urapidil].
Kardiologiia 52(1) , 86-90, (2012)
|
| 2,4(1H,3H)-Pyrimidinedione, 6-((3-(4-(2-methoxyphenyl)-1-piperazinyl)propyl)amino)-1,3-dimethyl-, monohydrochloride |
| 6-[3-[4-(2-methoxyphenyl)piperazin-1-yl]propylamino]-1,3-dimethylpyrimidine-2,4-dione,hydrochloride |
| 6-({3-[4-(2-Methoxyphenyl)piperazin-1-yl]propyl}amino)-1,3-dimethylpyrimidine-2,4(1H,3H)-dione hydrochloride (1:1) |
| 2,4(1H,3H)-Pyrimidinedione, 6-[[3-[4-(2-methoxyphenyl)-1-piperazinyl]propyl]amino]-1,3-dimethyl-, hydrochloride (1:1) |
| 6-({3-[4-(2-Methoxyphenyl)-1-piperazinyl]propyl}amino)-1,3-dimethyl-2,4(1H,3H)-pyrimidinedione hydrochloride (1:1) |