H-Tyr-OMe.HCl
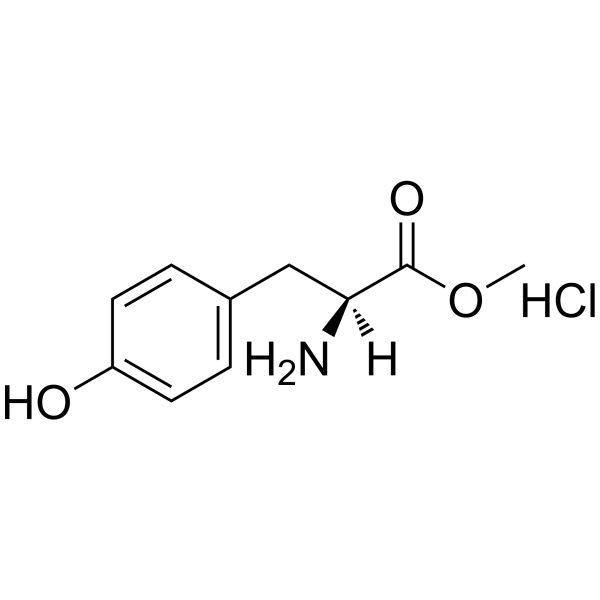
H-Tyr-OMe.HCl structure
|
Common Name | H-Tyr-OMe.HCl | ||
|---|---|---|---|---|
| CAS Number | 3417-91-2 | Molecular Weight | 231.676 | |
| Density | 1.21g/cm3 | Boiling Point | 330ºC at 760 mmHg | |
| Molecular Formula | C10H14ClNO3 | Melting Point | 192 °C (dec.)(lit.) | |
| MSDS | USA | Flash Point | 153.4ºC | |
Use of H-Tyr-OMe.HClH-Tyr-OMe.HCl is a tyrosine derivative[1]. |
| Name | Methyl L-tyrosinate hydrochloride |
|---|---|
| Synonym | More Synonyms |
| Description | H-Tyr-OMe.HCl is a tyrosine derivative[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Amino acids and amino acid derivatives have been commercially used as ergogenic supplements. They influence the secretion of anabolic hormones, supply of fuel during exercise, mental performance during stress related tasks and prevent exercise induced muscle damage. They are recognized to be beneficial as ergogenic dietary substances[1]. |
| References |
| Density | 1.21g/cm3 |
|---|---|
| Boiling Point | 330ºC at 760 mmHg |
| Melting Point | 192 °C (dec.)(lit.) |
| Molecular Formula | C10H14ClNO3 |
| Molecular Weight | 231.676 |
| Flash Point | 153.4ºC |
| Exact Mass | 231.066223 |
| PSA | 72.55000 |
| LogP | 1.93730 |
| Index of Refraction | 13 ° (C=2, MeOH) |
| InChIKey | VXYFARNRGZWHTJ-FVGYRXGTSA-N |
| SMILES | COC(=O)C(N)Cc1ccc(O)cc1.Cl |
| Storage condition | 2~8°C |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
|---|---|
| Hazard Codes | Xi:Irritant |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26-S36 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 29225000 |
| Precursor 10 | |
|---|---|
| DownStream 10 | |
| HS Code | 2922509090 |
|---|---|
| Summary | 2922509090. other amino-alcohol-phenols, amino-acid-phenols and other amino-compounds with oxygen function. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Transport and signaling via the amino acid binding site of the yeast Gap1 amino acid transceptor.
Nat. Chem. Biol. 5 , 45-52, (2009) Transporter-related nutrient sensors, called transceptors, mediate nutrient activation of signaling pathways through the plasma membrane. The mechanism of action of transporting and nontransporting tr... |
|
|
Novel molecular combination deriving from natural aminoacids and polyphenols: Design, synthesis and free-radical scavenging activities.
Eur. J. Med. Chem. 50 , 383-92, (2012) Following the recent output of scientific publications in the matter of synergic activity between different antioxidants, we have undertaken the present study with the aim to synthesize new molecules ... |
|
|
Aromatic amino acid methyl ester analogs form quinonoidal species with Dopa decarboxylase.
FEBS Lett. 412(1) , 245-8, (1997) This study reports for the first time that binding of aromatic methyl ester analogs to Dopa decarboxylase in the native and inactive nicked forms causes the appearance of a dead-end quinonoidal specie... |
| Methyl L-tyrosinate hydrochloride |
| L-Tyrosine Methylester HLC |
| L-TyrOCH3 hydrochloride |
| Tyr-OmeHCl |
| (S)-2-Amino-3-(4-hydroxyphenyl)propionic acid methyl ester hydrochloride |
| methyl (2S)-2-amino-3-(4-hydroxyphenyl)propanoate hydrochloride |
| METHYL TYROSINATE HCL |
| EINECS 222-313-3 |
| Tyrosine methyl ester hydrochloride, L- |
| L-Tyrosine Methyl Ester Hydrochloride |
| TYROSINE-OME HCL |
| L-TYROSINE METHYL ESTER HCL |
| L-Tyrosine, methyl ester, hydrochloride (1:1) |
| H-L-TYR-OME HCL |
| H-Tyr-OMe·HCl |
| (S)-Methyl 2-amino-3-(4-hydroxyphenyl)propanoate hydrochloride |
| L-Tyrosine, methyl ester, hydrochloride |
| L-Tyr-OMe.HCL |
| MethylL-tyrosinatehydrochloride |
| L-Tyr-OMe hydrochloride |
| MFCD00012607 |
| L-Tyr methyl ester hydrochloride |
| QR D1YZVO1 &&L Form HCl |
| L-Tyrosinemethylesterhydrochloride |
| Methyl L-tyrosinate hydrochloride (1:1) |
| H-Tyr-Ome HCl |
| H-Tyr-OMe.HCl |
 CAS#:67-56-1
CAS#:67-56-1 CAS#:60-18-4
CAS#:60-18-4 CAS#:13512-31-7
CAS#:13512-31-7 CAS#:3978-80-1
CAS#:3978-80-1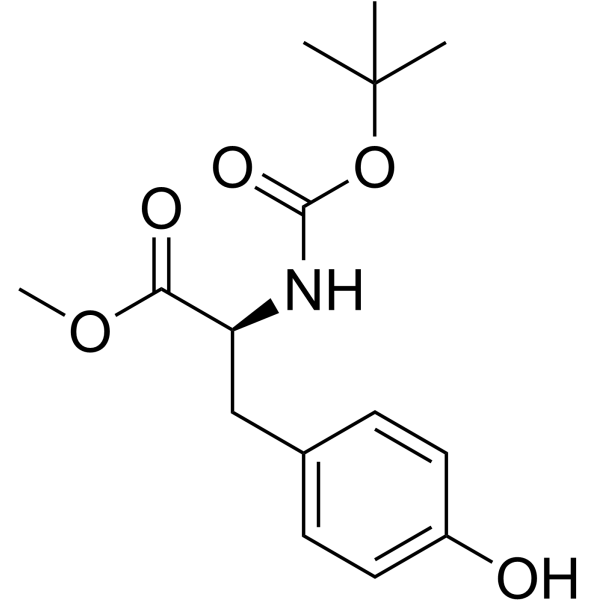 CAS#:4326-36-7
CAS#:4326-36-7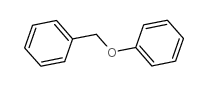 CAS#:946-80-5
CAS#:946-80-5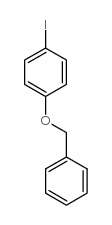 CAS#:19578-68-8
CAS#:19578-68-8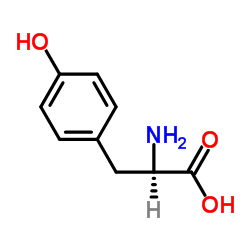 CAS#:556-02-5
CAS#:556-02-5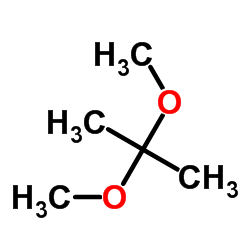 CAS#:77-76-9
CAS#:77-76-9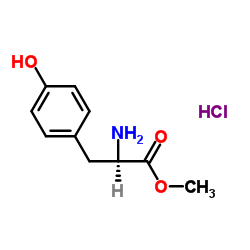 CAS#:3728-20-9
CAS#:3728-20-9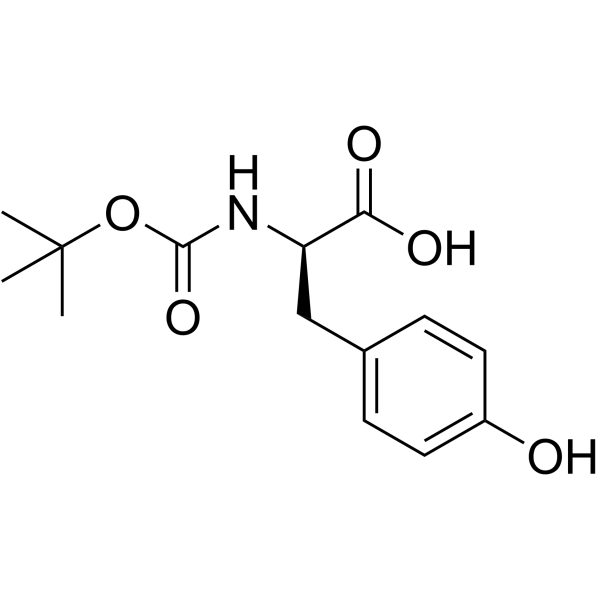 CAS#:70642-86-3
CAS#:70642-86-3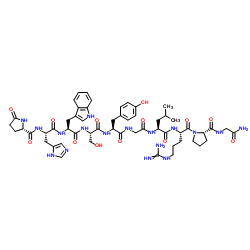 CAS#:33515-09-2
CAS#:33515-09-2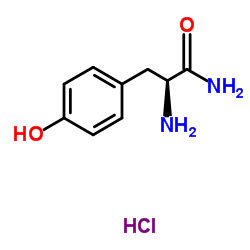 CAS#:4985-46-0
CAS#:4985-46-0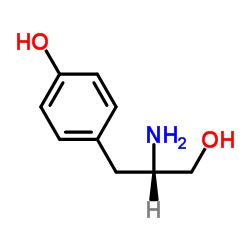 CAS#:5034-68-4
CAS#:5034-68-4![methyl 2-[(3-benzylsulfanyl-2-phenylmethoxycarbonylamino-propanoyl)amino]-3-(4-hydroxyphenyl)propanoate structure](https://image.chemsrc.com/caspic/310/19519-00-7.png) CAS#:19519-00-7
CAS#:19519-00-7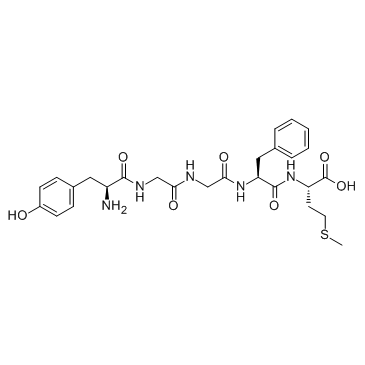 CAS#:58569-55-4
CAS#:58569-55-4
