spiraeoside
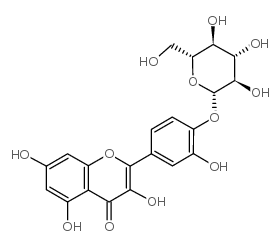
spiraeoside structure
|
Common Name | spiraeoside | ||
|---|---|---|---|---|
| CAS Number | 20229-56-5 | Molecular Weight | 464.37600 | |
| Density | 1.809g/cm3 | Boiling Point | 835.6ºC at 760mmHg | |
| Molecular Formula | C21H20O12 | Melting Point | 209-211ºC | |
| MSDS | Chinese USA | Flash Point | 294.6ºC | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of spiraeosideSpiraeoside, an orally active natural compound, exerts antioxidant activity, inhibits reactive oxygen species (ROS) and malondialdehyde production. Spiraeoside possesses antiallergic, anti-inflammatory and antitumor activities[1]. |
| Name | quercetin 4ʼ-O-β-D-glucopyranoside |
|---|---|
| Synonym | More Synonyms |
| Description | Spiraeoside, an orally active natural compound, exerts antioxidant activity, inhibits reactive oxygen species (ROS) and malondialdehyde production. Spiraeoside possesses antiallergic, anti-inflammatory and antitumor activities[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Spiraeoside elevates HG stimulation-caused the decrease in the expression levels of p-Akt, nuclear Nrf2, and HO-1 in AC16 cells (the effects of Spiraeoside are reversedby LY294002)[1]. Spiraeoside protects AC16 cells against HG-induced oxidative stress, cell injury, and apoptosis[1]. Spiraeoside activates the PI3K/Akt/Nrf2 pathway in AC16 cells on exposure to HG[1]. Spiraeoside protects AC16 cells against HG-induced apoptosis through the PI3K/Akt/Nrf2 pathway[1]. Cell Viability Assay[1] Cell Line: AC16 cells. Concentration: 1, 5, 10, or 20 μM. Incubation Time: 0, 24, or 48 hours. Result: Inhibited AC16 cells viability (20 μM). |
| In Vivo | Spiraeoside (50 mg/kg, p.o.) shows ulcer preventive ability[2]. Animal Model: Male Wistar rats (6-8 weeks old)[2]. Dosage: 50 mg/kg. Administration: Oral gavage an hour before inducing the lesions. Result: Decreased severity of the formed lesions. |
| References |
| Density | 1.809g/cm3 |
|---|---|
| Boiling Point | 835.6ºC at 760mmHg |
| Melting Point | 209-211ºC |
| Molecular Formula | C21H20O12 |
| Molecular Weight | 464.37600 |
| Flash Point | 294.6ºC |
| Exact Mass | 464.09500 |
| PSA | 210.51000 |
| Vapour Pressure | 1.28E-29mmHg at 25°C |
| Index of Refraction | 1.774 |
| Storage condition | ?20°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATAMUTATION DATA
|
|
The crystal structure of human cytosolic beta-glucosidase unravels the substrate aglycone specificity of a family 1 glycoside hydrolase.
J. Mol. Biol. 370(5) , 964-75, (2007) Human cytosolic beta-glucosidase (hCBG) is a xenobiotic-metabolizing enzyme that hydrolyses certain flavonoid glucosides, with specificity depending on the aglycone moiety, the type of sugar and the l... |
|
|
Flavonoid glycosides inhibit oral cancer cell proliferation--role of cellular uptake and hydrolysis to the aglycones.
J. Pharm. Pharmacol. 57(8) , 1037-42, (2005) Epidemiologic evidence supports the view that dietary flavonoids exert protective effects in oral diseases, including cancer. However, the dietary forms of flavonoids, the flavonoid glycosides, are th... |
|
|
Deglycosylation of flavonoid and isoflavonoid glycosides by human small intestine and liver beta-glucosidase activity.
FEBS Lett. 436(1) , 71-5, (1998) Flavonoid and isoflavonoid glycosides are common dietary phenolics which may be absorbed from the small intestine of humans. The ability of cell-free extracts from human small intestine and liver to d... |
| Spiraeosid |
| Quercetin 4'-b-D-glucopyranoside |
| Quercetin 4'-O-glucoside |
| Quercetin-4'-glucoside |
| EINECS 243-614-6 |
| Spiraein |
| 3,5,7-trihydroxy-2-[3-hydroxy-4-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxyphenyl]chromen-4-one |
| quercetin 4'-O-beta-D-glucopyranoside |
| Spiraeoside |
 CAS#:1313191-85-3
CAS#:1313191-85-3 CAS#:117-39-5
CAS#:117-39-5 CAS#:572-09-8
CAS#:572-09-8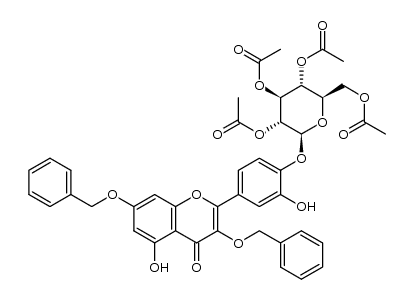 CAS#:1313191-83-1
CAS#:1313191-83-1![2-(2,2-diphenylbenzo[d][1,3]dioxol-5-yl)-3,5,7-trihydroxy-4H-chroMen-4-one Structure](https://image.chemsrc.com/caspic/359/357194-03-7.png) CAS#:357194-03-7
CAS#:357194-03-7 CAS#:133-89-1
CAS#:133-89-1 CAS#:2280-44-6
CAS#:2280-44-6
