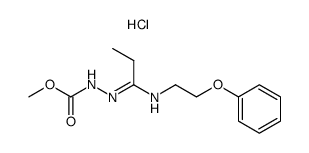
83366-66-9
| Name | nefazodone |
|---|---|
| Synonyms |
[14C]-Nefazodone hydrochloride
Nefazodonum Nefazodonum [Latin] 2-[3-[4-(3-chlorophenyl)-1-piperazinyl]-propyl-]5-ethyl-2,4-dihydro-4-(2-phenoxy-ethyl)-3H-1,2,4-triazol-3-one 2-<3-<4-(3-chlorophenyl)-1-piperazinyl>propyl>-5-ethyl-2,4-dihydro-4-(2-phenoxyethyl)-3H-1,2,4-triazolin-3-one Nefazodone Hcl 2-[3-[4-(3-chlorophenyl)piperazin-1-yl]propyl]-5-ethyl-4-(2-phenoxyethyl)-1,2,4-triazol-3-one Nefazodona Nefazodone (INN) Nefazodona [Spanish] 2-[3-[4-(3-chlorophenyl)-1-piperazinyl]propyl]-5-ethyl-4-(2-phenoxyethyl)-2H-1,2,4-triazol-3(4H)-one UNII-59H4FCV1TF MFCD00935760 |
| Description | Nefazodone is an orally active phenylpiperazine antidepressant. Nefazodone can potently and selectively block postsynaptic 5-HT2A receptors, and moderately inhibit 5-HT and noradrenaline reuptake. Nefazodone can also relieve the adverse effects of stress on the the immune system of mice. Nefazodone has a high affinity for CYP3A4 isoenzyme, which indicates that it has certain risk of drug-drug interaction[1][2][3]. |
|---|---|
| Related Catalog | |
| Target |
5-HT receptor, Noradrenaline, CYP3A4[1] |
| In Vitro | Nefazodone collapses mitochondrial membrane potential, and imposes oxidative stress, as detected via glutathione depletion, leading to cell death[2]. Nefazodone (200 μM; 24 h) depletes 100% of ATP in both, glucose and galactose-grown HepG2 cells[2]. Nefazodone (6.25, 12.5 and 25 μM; 0-120 min) profoundly inhibits oxygen consumption (OCR) in HepG2[2]. |
| In Vivo | Nefazodone (10 mg/kg; s.c.; for 16 days) is effective to counter the adverse effects of stress on the the immune system of mice[3]. Animal Model: Female BALB/c mice (7-12 weeks old; stress model; subjected to a broad band noise at 100 dB daily for 5 s every minute during a 1- or 3-h period around midnight)[3] Dosage: 10 mg/kg Administration: s.c.; for 16 days Result: Attenuated the reduction of thymus, spleen and peripheral blood cellularity caused by stress. |
| Melting Point | 180-182°C |
|---|---|
| Molecular Formula | C25H32ClN5O2 |
| Molecular Weight | 470.00700 |
| Exact Mass | 469.22400 |
| PSA | 55.53000 |
| LogP | 3.55480 |
| Water Solubility | DMSO: ~11 mg/mL at 60 °C |
| Safety Phrases | S22-S24/25 |
|---|---|
| WGK Germany | 3 |
|
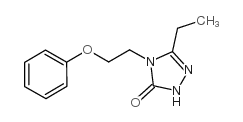
~82% 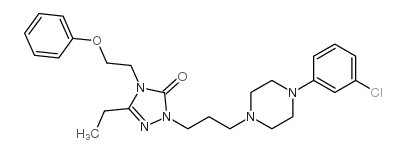
83366-66-9 |
| Literature: Madding; Smith; Sheldon; Lee Journal of Heterocyclic Chemistry, 1985 , vol. 22, # 4 p. 1121 - 1126 |
|
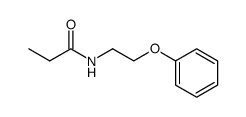
~% 
83366-66-9 |
| Literature: Journal of Heterocyclic Chemistry, , vol. 22, # 4 p. 1121 - 1126 |
|
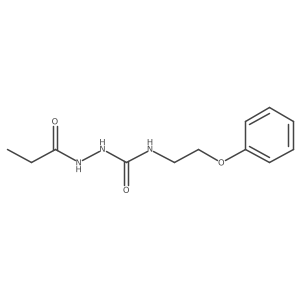
~% 
83366-66-9 |
| Literature: Journal of Heterocyclic Chemistry, , vol. 22, # 4 p. 1121 - 1126 |
|
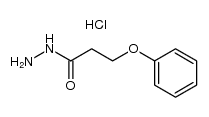
~% 
83366-66-9 |
| Literature: Journal of Heterocyclic Chemistry, , vol. 22, # 4 p. 1121 - 1126 |
|
~% 
83366-66-9 |
| Literature: Journal of Heterocyclic Chemistry, , vol. 22, # 4 p. 1121 - 1126 |
|
~% 
83366-66-9 |
| Literature: Journal of Heterocyclic Chemistry, , vol. 22, # 4 p. 1121 - 1126 |
| Precursor 4 | |
|---|---|
| DownStream 0 | |