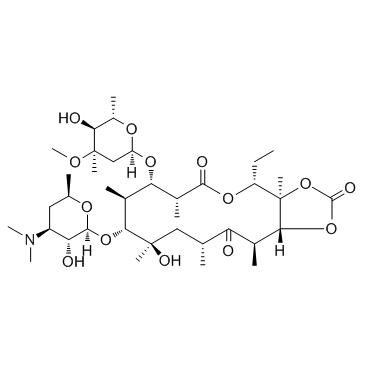
55224-05-0
| Name | Davercin |
|---|---|
| Synonyms |
Erythromycin Cyclocarbonate
CWE Erythromycin Cyclocarbonate Dravercin Eritromicina A carbonato Carbonato ciclico de eritromicina a Cykliczny weglan erytromycyny a Cykliczny weglan erytromycyny A [polish] erythromycin A cyclic 11,12-carbonate Eritromicina A carbonato [spanish] |
| Description | Erythromycin Cyclocarbonate, derivative of Erythromycin, which is active against Gram-positive and some Gram-negative microorganisms. |
|---|---|
| Related Catalog | |
| Target |
Antibacterial[1] |
| In Vitro | Erythromycin is used in treatment of respiratory, gastrointestinal, and genital tract infections, as well as skin and soft tissue infections. Erythromycin, with its ten chiral centers and two sugar substituents (L-cladinose and D-desosamine), is a good starting point for numerous medicinal chemistry efforts for improvement of its biological profile (better activity, higher stability, and improved bioavailability) since the first generation of macrolides, which had low toxicity and good tolerability, are unstable in acidic media, had low toxicity and good tolerability[1]. |
| References |
| Density | 1.22 g/cm3 |
|---|---|
| Boiling Point | 859.8ºC at 760 mmHg |
| Molecular Formula | C38H65NO14 |
| Molecular Weight | 759.92100 |
| Exact Mass | 759.44100 |
| PSA | 188.98000 |
| LogP | 2.96780 |
| Index of Refraction | 1.534 |
| Storage condition | -20°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|