552-59-0
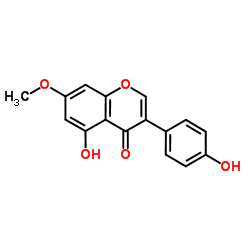
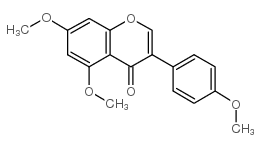
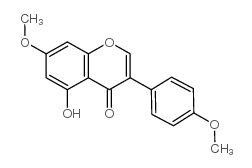
| Name | prunetin |
|---|---|
| Synonyms |
5-Hydroxy-3-(4-hydroxyphenyl)-7-methoxy-4H-chromen-4-one
Prunusetin 5,4'-dihydroxy-7-methoxyisoflavone Prunetin 7-methoxy-4',5-dihydroxyisoflavone 5-hydroxy-3-(4-hydroxyphenyl)-7-methoxychromen-4-one 7-O-methyl-genistein 4',5-dihydroxy-7-methoxyisoflavone 4',5-dihydroxy-7-methoxygenistein EINECS 209-018-5 MFCD00016951 |
| Description | Prunetin, an O-methylated isoflavone, possesses anti-inflammatory activity. Prunetin is a potent human aldehyde dehydrogenases inhibitor[1][2]. |
|---|---|
| Related Catalog | |
| In Vitro | Prunetin inhibited LPS-induced inflammatory cytokine production and MUC5 AC expression and secretion by inactivating the TLR4/MyD88 pathway in human nasal epithelial cells[1]. |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 546.5±50.0 °C at 760 mmHg |
| Melting Point | 240-242ºC |
| Molecular Formula | C16H12O5 |
| Molecular Weight | 284.263 |
| Flash Point | 209.7±23.6 °C |
| Exact Mass | 284.068481 |
| PSA | 79.90000 |
| LogP | 3.53 |
| Vapour Pressure | 0.0±1.5 mmHg at 25°C |
| Index of Refraction | 1.669 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi |
| Safety Phrases | S22-S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | DJ3100050 |
| HS Code | 2914509090 |
| Precursor 9 | |
|---|---|
| DownStream 2 | |
| HS Code | 2914509090 |
|---|---|
| Summary | HS:2914509090 other ketones with other oxygen function VAT:17.0% Tax rebate rate:9.0% Supervision conditions:none MFN tariff:5.5% General tariff:30.0% |