518-69-4
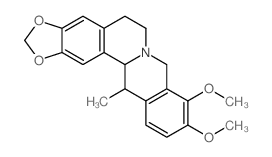
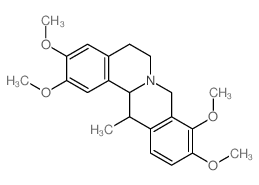
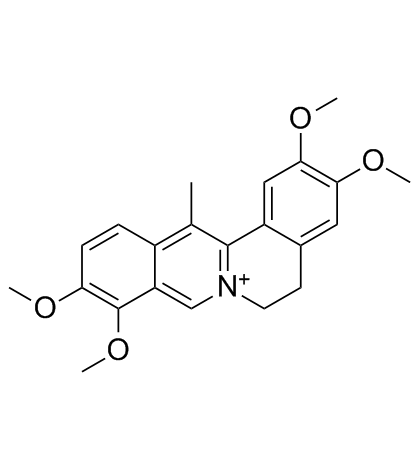
| Name | corydaline |
|---|---|
| Synonyms |
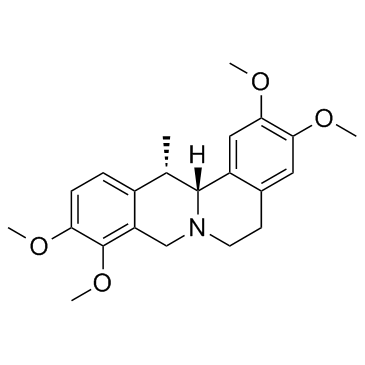
(13S,13aR)-2,3,9,10-tetramethoxy-13-methyl-6,8,13,13a-tetrahydro-5H-isoquinolino[2,1-b]isoquinoline
d-Corydalin (13S,13aR)-2,3,9,10-Tetramethoxy-13-methyl-5,8,13,13a-tetrahydro-6H-isoquinolino[3,2-a]isoquinoline (13S-trans )-5,8,13,13a-Tetrahydro-2,3,9,10-tetramethoxy-13-methyl-6H-dibenzo[a,g]quinolizine Corydaline (+) Berbine,2,3,9,10-tetramethoxy-13-methyl 2,3,9,10-Tetramethoxy-13a-methyl-13ab-berbine (13S,13aR)-2,3,9,10-Tetramethoxy-13-methyl-5,8,13,13a-tetrahydro-6H-isoquino[3,2-a]isoquinoline Corydalin Corydaline |
| Description | Corydaline is an acetylcholinesterase inhibitor isolated from Corydalis yanhusuo. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 482.3±45.0 °C at 760 mmHg |
| Melting Point | 135° |
| Molecular Formula | C22H27NO4 |
| Molecular Weight | 369.454 |
| Flash Point | 135.7±25.9 °C |
| Exact Mass | 369.194000 |
| PSA | 40.16000 |
| LogP | 4.19 |
| Vapour Pressure | 0.0±1.2 mmHg at 25°C |
| Index of Refraction | 1.600 |
| Storage condition | -20°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
|
~% 
518-69-4 |
| Literature: Manske Journal of the American Chemical Society, 1953 , vol. 75, p. 4928 |
|
~% 
518-69-4 |
| Literature: CVI PHARMACEUTICALS LIMITED; LIU, Haiyan; LI, Gaoping; WANG, Junbo; LIU, Jingwen Patent: WO2010/75469 A1, 2010 ; Location in patent: Page/Page column 62 ; WO 2010/075469 A1 |
|
~% 
518-69-4 |
| Literature: Dobbie; Lauder; Paliatseas Journal of the Chemical Society, 1901 , vol. 79, p. 89 |
|
~% 
518-69-4 |
| Literature: Gadamer; Wagner Archiv der Pharmazie (Weinheim, Germany), 1902 , vol. 240, p. 36 |
|
~% 
518-69-4 |
| Literature: Gadamer; Wagner Archiv der Pharmazie (Weinheim, Germany), 1902 , vol. 240, p. 36 |
| Precursor 5 | |
|---|---|
| DownStream 3 | |