H-Phe-ol
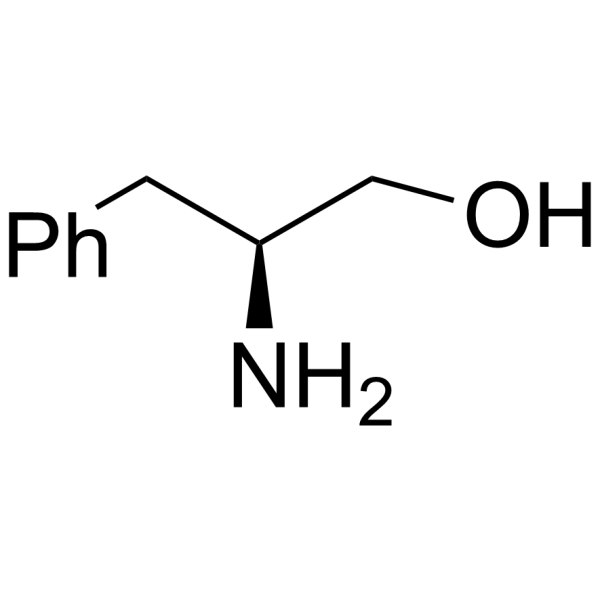
H-Phe-ol structure
|
Common Name | H-Phe-ol | ||
|---|---|---|---|---|
| CAS Number | 3182-95-4 | Molecular Weight | 151.206 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 303.8±22.0 °C at 760 mmHg | |
| Molecular Formula | C9H13NO | Melting Point | 92-94ºC | |
| MSDS | Chinese USA | Flash Point | 137.5±22.3 °C | |
| Symbol |

GHS05 |
Signal Word | Danger | |
Use of H-Phe-olL-(-)-Phenylalaninol is an alanine derivative[1]. |
| Name | (2S)-2-amino-3-phenylpropan-1-ol |
|---|---|
| Synonym | More Synonyms |
| Description | L-(-)-Phenylalaninol is an alanine derivative[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Amino acids and amino acid derivatives have been commercially used as ergogenic supplements. They influence the secretion of anabolic hormones, supply of fuel during exercise, mental performance during stress related tasks and prevent exercise induced muscle damage. They are recognized to be beneficial as ergogenic dietary substances[1]. |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 303.8±22.0 °C at 760 mmHg |
| Melting Point | 92-94ºC |
| Molecular Formula | C9H13NO |
| Molecular Weight | 151.206 |
| Flash Point | 137.5±22.3 °C |
| Exact Mass | 151.099716 |
| PSA | 46.25000 |
| LogP | 0.77 |
| Vapour Pressure | 0.0±0.7 mmHg at 25°C |
| Index of Refraction | 1.561 |
| InChIKey | STVVMTBJNDTZBF-VIFPVBQESA-N |
| SMILES | NC(CO)Cc1ccccc1 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS05 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H314 |
| Precautionary Statements | P280-P305 + P351 + P338-P310 |
| Personal Protective Equipment | Eyeshields;Faceshields;full-face particle respirator type N100 (US);Gloves;respirator cartridge type N100 (US);type P1 (EN143) respirator filter;type P3 (EN 143) respirator cartridges |
| Hazard Codes | C: Corrosive; |
| Risk Phrases | R34 |
| Safety Phrases | S24/25 |
| RIDADR | UN 3259 8/PG 3 |
| WGK Germany | 3 |
| RTECS | UA6900000 |
| Hazard Class | 8.0 |
| HS Code | 29221980 |
| Precursor 9 | |
|---|---|
| DownStream 10 | |
| HS Code | 2922199090 |
|---|---|
| Summary | 2922199090. other amino-alcohols, other than those containing more than one kind of oxygen function, their ethers and esters; salts thereof. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Straightforward methodology for the enantioselective synthesis of benzo[a]- and indolo[2,3-a]quinolizidines.
J. Org. Chem. 72(14) , 5193-201, (2007) An enantioselective two-step route to substituted benzo[a]- and indolo[2,3-a]quinolizidines has been developed. It consists of (i) a stereoselective cyclocondensation of a racemic or prochiral delta-o... |
|
|
Direct high-performance liquid chromatographic separation of the enantiomers of an aromatic amine and four aminoalcohols using polysaccharide chiral stationary phases and acidic additive.
Chirality 19(8) , 647-53, (2007) The HPLC enantiomeric separation of N-benzyl-alpha-methyl-benzylamine, phenylalaninol, tryptophanol, 2 (diphenylhydroxymethyl)pyrrolidine, and isoproterenol was accomplished in the normal-phase mode u... |
|
|
Paracelsin; characterization by NMR spectroscopy and circular dichroism, and hemolytic properties of a peptaibol antibiotic from the cellulolytically active mold Trichoderma reesei. Part B.
Experientia 40 , 1189, (1984) Paracelsin, a hemolytic and membrane active polypeptide antibiotic of the peptaibol class which is excreted by the mold Trichoderma reesei, was obtained by a simplified and rapid isolation procedure u... |
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: qHTS for Inhibitors of human tyrosyl-DNA phosphodiesterase 1 (TDP1): qHTS in cells in...
Source: NCGC
Target: TDP1 protein [Homo sapiens]
External Id: TDP1100
|
|
Name: qHTS for Inhibitors of human tyrosyl-DNA phosphodiesterase 1 (TDP1): qHTS in cells in...
Source: NCGC
Target: TDP1 protein [Homo sapiens]
External Id: TDP1101
|
|
Name: Schnurri-3 Inhibitors: specific inducers of adult bone formation Measured in Cell-Bas...
Source: Broad Institute
Target: N/A
External Id: 2134-01_Inhibitor_SinglePoint_HTS_Activity_Set2
|
|
Name: Spectrum HTS for Inhibitors of Aerobactin Synthetase IucA
Source: 23265
Target: IucA Synthetase from hypervirulent Klebsiella pneumoniae hvKP1
External Id: IucA Pilot Assay Spectrum Library
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
|
Name: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfect...
Source: Broad Institute
Target: N/A
External Id: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfected HEK293 cells Inhibition - 7011-01_Antagonist_SinglePoint_HTS_Activity
|
|
Name: The chemical genetic matrix (CGM) dataset as reported in Wildenhain et al. (2015) Pre...
Source: 11924
Target: N/A
External Id: CGM data for Cell Systems paper Dec 2015
|
| β-Anilinoethanol |
| HPH |
| (S)-(-)-2-amino-3-phenylpropan-1-ol |
| (S)-2-Amino-3-phenyl-1-propanol |
| (S)-2-Amino-3-phenylpropan-1-ol |
| (S)-2-amino-2-phenylpropan-1-ol |
| (S)-2-Amino-3-phenyl-propan-1-ol |
| N-(β-hydroxy)ethylaniline |
| L(-)-2-Amino-3-phenyl-1-propanol |
| 2-(phenylamino)ethanol |
| EINECS 221-674-4 |
| H-Phenylalaninol |
| (S)-(-)-2-Amino-3-phenyl-1-propanol |
| (S)-(-)-phenylalaninol |
| (2S)-2-amino-3-phenylpropanol |
| (2S)-2-Amino-3-phenyl-1-propanol |
| (-)-L-PHENYLALANINOL |
| (2S)-2-amino-3-phenylpropan-1-ol |
| L-phenylalaninol |
| (S)-β-Aminobenzenepropanol |
| phenylalaninol |
| 2-amino-3-phenylpropan-1-ol |
| (S)-(−)-2-Amino-3-phenyl-1-propanol |
| N-(β-Hydroxyethyl)aniline |
| 2-Amino-3-Phenyl-Propan-1-Ol |
| 2-Anilinoethanol |
| H-Phe-ol |
| MFCD00004732 |
| (±)-phenylethanolamine |
| N-2-hydroxyethylaniline |
| L-Phenylglycinol |
 CAS#:63-91-2
CAS#:63-91-2 CAS#:7524-50-7
CAS#:7524-50-7 CAS#:2577-90-4
CAS#:2577-90-4 CAS#:6372-14-1
CAS#:6372-14-1 CAS#:3182-93-2
CAS#:3182-93-2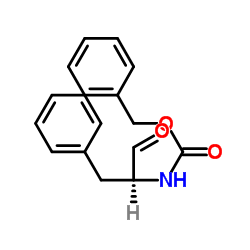 CAS#:59830-60-3
CAS#:59830-60-3 CAS#:150-30-1
CAS#:150-30-1 CAS#:2935-35-5
CAS#:2935-35-5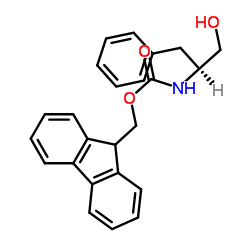 CAS#:129397-83-7
CAS#:129397-83-7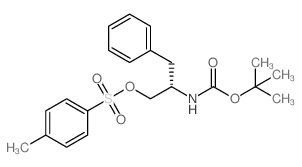 CAS#:141403-49-8
CAS#:141403-49-8![1-[(2S)-1-hydroxy-3-phenylpropan-2-yl]-3-phenylurea structure](https://image.chemsrc.com/caspic/384/223600-53-1.png) CAS#:223600-53-1
CAS#:223600-53-1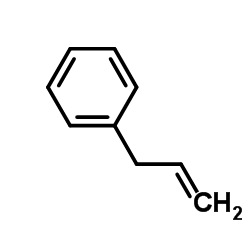 CAS#:300-57-2
CAS#:300-57-2 CAS#:93-54-9
CAS#:93-54-9 CAS#:1334326-63-4
CAS#:1334326-63-4 CAS#:29802-26-4
CAS#:29802-26-4 CAS#:82495-70-3
CAS#:82495-70-3 CAS#:90719-32-7
CAS#:90719-32-7 CAS#:2448-45-5
CAS#:2448-45-5![1-[4-[2-[(4-chlorophenyl)-phenylmethoxy]ethyl]piperazin-1-yl]propan-2-ol,oxalic acid structure](https://image.chemsrc.com/caspic/355/2323-36-6.png) CAS#:2323-36-6
CAS#:2323-36-6
