| Structure | Name/CAS No. | Articles |
|---|---|---|
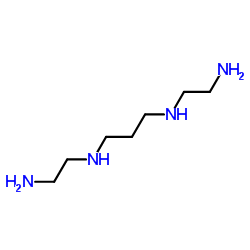 |
n,n'-bis(2-aminoethyl)propan-1,3-diamin
CAS:4741-99-5 |
|
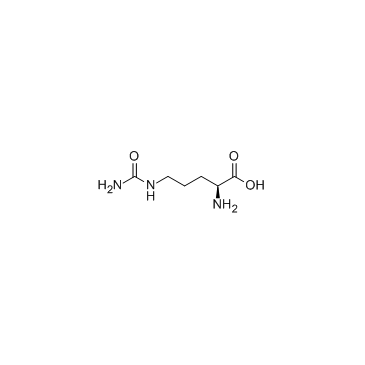 |
L-Citruline
CAS:372-75-8 |
|
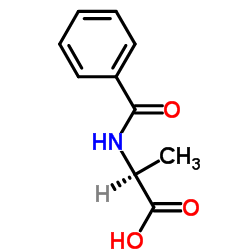 |
BZ-ALA-OH
CAS:2198-64-3 |
|
 |
Protein-arginine deiminase
CAS:75536-80-0 |