| Structure | Name/CAS No. | Articles |
|---|---|---|
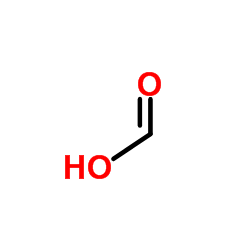 |
Formic Acid
CAS:64-18-6 |
|
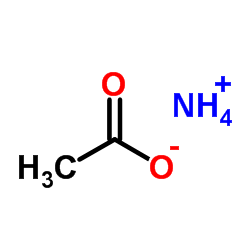 |
Ammonium acetate
CAS:631-61-8 |
|
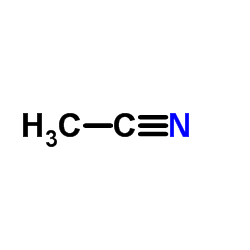 |
Acetonitrile
CAS:75-05-8 |
|
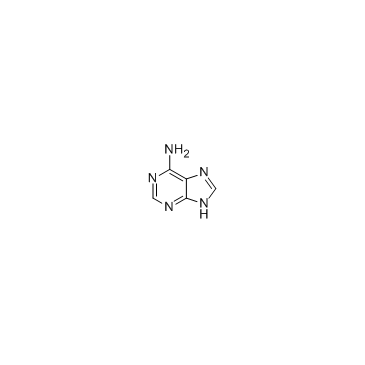 |
Adenine
CAS:73-24-5 |
|
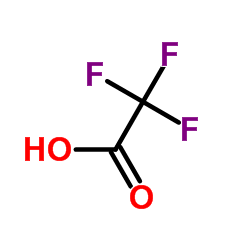 |
trifluoroacetic acid
CAS:76-05-1 |
|
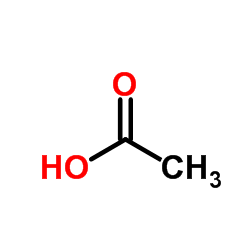 |
acetic acid
CAS:64-19-7 |
|
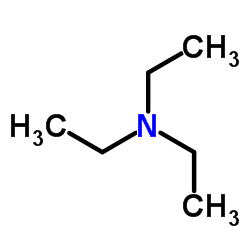 |
Triethylamine
CAS:121-44-8 |
|
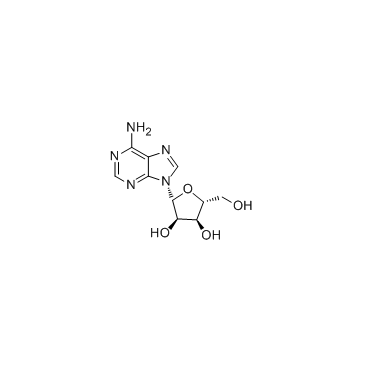 |
Adenosine
CAS:58-61-7 |
|
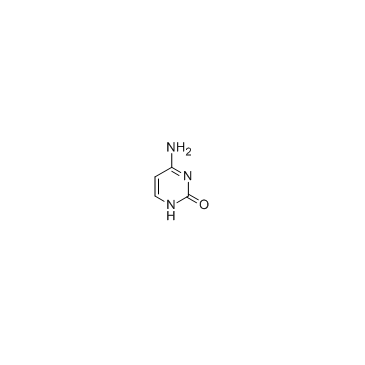 |
cytosine
CAS:71-30-7 |
|
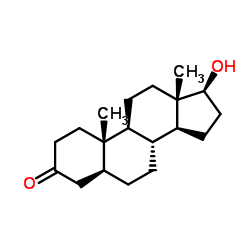 |
Stanolone
CAS:521-18-6 |