Vigabatrin
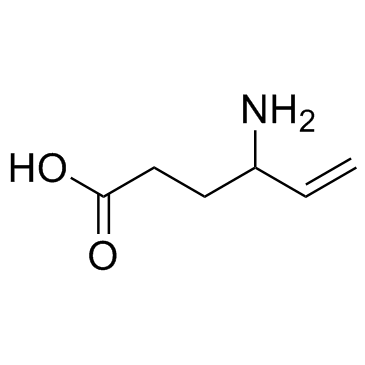
Vigabatrin structure
|
Common Name | Vigabatrin | ||
|---|---|---|---|---|
| CAS Number | 68506-86-5 | Molecular Weight | 129.157 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 277.7±28.0 °C at 760 mmHg | |
| Molecular Formula | C6H11NO2 | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 121.7±24.0 °C | |
|
Cheminformatics analysis of assertions mined from literature that describe drug-induced liver injury in different species.
Chem. Res. Toxicol. 23 , 171-83, (2010) Drug-induced liver injury is one of the main causes of drug attrition. The ability to predict the liver effects of drug candidates from their chemical structures is critical to help guide experimental drug discovery projects toward safer medicines. In this st... |
|
|
Translating clinical findings into knowledge in drug safety evaluation--drug induced liver injury prediction system (DILIps).
J. Sci. Ind. Res. 65(10) , 808, (2006) Drug-induced liver injury (DILI) is a significant concern in drug development due to the poor concordance between preclinical and clinical findings of liver toxicity. We hypothesized that the DILI types (hepatotoxic side effects) seen in the clinic can be tra... |
|
|
Chemical genetics reveals a complex functional ground state of neural stem cells.
Nat. Chem. Biol. 3(5) , 268-273, (2007) The identification of self-renewing and multipotent neural stem cells (NSCs) in the mammalian brain holds promise for the treatment of neurological diseases and has yielded new insight into brain cancer. However, the complete repertoire of signaling pathways ... |
|
|
Neural computational prediction of oral drug absorption based on CODES 2D descriptors.
Eur. J. Med. Chem. 45 , 930-40, (2010) A neural model based on a numerical molecular representation using CODES program to predict oral absorption of any structure is described. This model predicts both high and low-absorbed compounds with a global accuracy level of 74%. CODES/ANN methodology show... |
|
|
Experimental solubility profiling of marketed CNS drugs, exploring solubility limit of CNS discovery candidate.
Bioorg. Med. Chem. Lett. 20 , 7312-6, (2010) We determined the experimental solubility of CNS marketed drugs. Of the 98 drugs measured, greater than 90% had solubility >10 μM in pH 7.4 buffer. Only seven drugs had solubility <10 μM. Using these data, we established a solubility criterion to support CNS ... |
|
|
A multisite, double-blind, placebo-controlled clinical trial to evaluate the safety and efficacy of vigabatrin for treating cocaine dependence.
JAMA Psychiatry 70(6) , 630-7, (2013) Cocaine dependence is a significant public health problem, yet no validated pharmacological treatment exists. The potent γ-aminobutyric acid (GABA)ergic medication vigabatrin has previously been shown to be effective in a double-blind single-site study conduc... |
|
|
Cerebellar fastigial nuclear GABAergic projections to the hypothalamus modulate immune function.
Brain. Behav. Immun. 27(1) , 80-90, (2013) Our previous work has shown that the cerebellar fastigial nucleus (FN) is involved in modulation of lymphocyte function. Herein, we investigated effect of FN γ-aminobutyric acid (GABA)-ergic projections to the hypothalamus on lymphocytes to understand pathway... |
|
|
Vigabatrin versus carbamazepine monotherapy for epilepsy.
Cochrane Database Syst. Rev. 1 , CD008781, (2012) The efficacy and safety of vigabatrin (VGB) as an add-on therapy for refractory epilepsy has been well established. However, this needs to be weighed against the risk of the development of visual field defects. Whether VGB monotherapy is an effective and safe... |
|
|
Changes in the ERG d-wave with vigabatrin treatment in a pediatric cohort.
Doc. Ophthalmol. 129(2) , 97-104, (2014) Vigabatrin (VGB), a treatment for the childhood epilepsy, infantile spasms (IS), is implicated in visual field constriction. Electroretinograms (ERGs) are used as a substitute for visual field testing in infants. We use the VGB-associated ERG reduction (VAER)... |
|
|
Vigabatrin-associated retinal damage: potential biochemical mechanisms.
Acta Neurol. Scand 126(4) , 219-28, (2012) Vigabatrin (VGB), an irreversible inhibitor of gamma-aminobutyric acid (GABA) transaminase, is approved as adjunct treatment of refractory partial seizures as well as infantile spasms. Although VGB has been proven to be effective, its use is limited by the ri... |